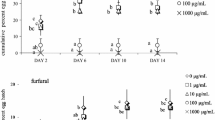
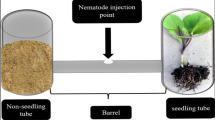
Abstract
Nematodes from the genus Meloidogyne are extremely harmful to agricultural crops due to their wide geographic distribution, variety of hosts, and strong reproductive capacity. The most used phytonematoid control method is the application of nematicides, which can also affect human health and the environment. Selenol ester derivatives have pharmaceutical applications, but little is known about their biological activities. We examined whether six selenol ester derivatives were nematicidal and ovicidal to the nematodes Caenorhabditis elegans and Meloidogyne incognita, and determined their toxicity to non-target organisms. The compound Se-p-methyl phenylselenobenzoate (2e) exhibited the lowest nematicidal and ovicidal Lethal concentration (LC50) to C. elegans (4.42 and 0.56 mg/L, respectively) and M. incognita (4.6 and 2.77 mg/L, respectively), which were similar or lower than the LC50 values for the control treatment with fluensulfone in C. elegans (4.07 and 6.05 mg/L, respectively) and M. incognita (43.07 and 49.63 mg/L, respectively). Compound 2e efficiently controlled the phytoparasite in greenhouse and reduced the number of galls and eggs present in tomato roots. Compared with the negative control, compound 2e was not toxic to other organisms such as Chlorella vulgaris algae, Galleria mellonella and Aedes aegypti larvae, and the aquatic protozoan Tetrahymena pyriformis. Our findings indicate that the selenol ester derivatives are potent and effective nematicides with no toxicity to the non-target organisms tested.





Similar content being viewed by others
References
Abd-Elgawad MMM, Askary TH (2015) Impact of phytonematodes on agriculture economy. Biocontrol agents of phytonematodes. CABI, Wallingford, pp 3–49
Álvarez-Pérez M, Ali W, Marć M et al (2018) Selenides and diselenides: a review of their anticancer and chemopreventive activity. Molecules 23:628. https://doi.org/10.3390/molecules23030628
APVMA (2015) Public release summary on the evaluation of the New Active Fluensulfone
Barbosa FAR, Canto RFS, Saba S et al (2016) Synthesis and evaluation of dihydropyrimidinone-derived selenoesters as multi-targeted directed compounds against Alzheimer’s disease. Bioorg Med Chem 24:5762–5770. https://doi.org/10.1016/j.bmc.2016.09.031
Barry KH, Koutros S, Lubin JH et al (2012) Methyl bromide exposure and cancer risk in the Agricultural Health Study. Cancer Causes Control 23:807–818. https://doi.org/10.1007/s10552-012-9949-2
Beletskaya IP, Sigeev AS, Peregudov AS, Petrovskii PV (2001) Tributylstannyl aryl selenides as efficient arylselenating agents in the synthesis of seleno esters. Russ J Org Chem. https://doi.org/10.1023/A:1013913816069
Bernard GC, Egnin M, Bonsi C (2017) The impact of plant-parasitic nematodes on agriculture and methods of control. In: Nematology—concepts, diagnosis and control. InTech. https://doi.org/10.5772/intechopen.68958
Bi Z, Gong Y, Huang X et al (2015) Efficacy of four nematicides against the reproduction and development of pinewood Nematode, Bursaphelenchus xylophilus. J Nematol 47(2):126–132. http://www.ncbi.nlm.nih.gov/pubmed/26170474
Bonetti JI, Ferraz S (1981) Modificações do método de Hussey & Barker para extração de ovos de Meloidogyne exigua em raízes de cafeeiro. Fitopatol Bras 6:553
Boyd WA, Smith MV, Freedman JH (2012) Caenorhabditis elegans as a model in developmental toxicology. In: Methods in molecular biology, pp 15–24. https://doi.org/10.1007/978-1-61779-867-2_3
Budnik LT, Kloth S, Velasco-Garrido M, Baur X (2012) Prostate cancer and toxicity from critical use exemptions of methyl bromide: environmental protection helps protect against human health risks. Environ Health 11:5. https://doi.org/10.1186/1476-069X-11-5
Calma ML, Medina PMB (2020) Acute and chronic exposure of the holometabolous life cycle of Aedes aegypti L to emerging contaminants naproxen and propylparaben. Environ Pollut 266:115275. https://doi.org/10.1016/j.envpol.2020.115275
Che Z, Zhang S, Shao Y et al (2013) Synthesis and quantitative structure-activity relationship (QSAR) study of novel N -arylsulfonyl-3-acylindole arylcarbonyl hydrazone derivatives as nematicidal agents. J Agric Food Chem 61:5696–5705. https://doi.org/10.1021/jf400536q
Chen J, Wei C, Wu S et al (2020) Novel 1,3,4-oxadiazole thioether derivatives containing flexible-chain moiety: design, synthesis, nematocidal activities, and pesticide-likeness analysis. Bioorg Med Chem Lett 30:127028. https://doi.org/10.1016/j.bmcl.2020.127028
Circu ML, Aw TY (2010) Reactive oxygen species, cellular redox systems, and apoptosis. Free Radic Biol Med 48:749–762. https://doi.org/10.1016/j.freeradbiomed.2009.12.022
DE Pires V, Blundell TL, Ascher DB (2015) pkCSM: predicting small-molecule pharmacokinetic and toxicity properties using graph-based signatures. J Med Chem 58:4066–4072. https://doi.org/10.1021/acs.jmedchem.5b00104
de Souza A, Narvencar KPS, Sindhoora KV (2013) The neurological effects of methyl bromide intoxication. J Neurol Sci 335:36–41. https://doi.org/10.1016/j.jns.2013.09.022
Desjardins D, Cacho-Valadez B, Liu J et al (2017) Antioxidants reveal an inverted U-shaped dose-response relationship between reactive oxygen species levels and the rate of aging in Caenorhabditis elegans. Aging Cell 16:104–112. https://doi.org/10.1111/acel.12528
DeLorenzo ME, Scott GI, Ross PE (2001) Toxicity of pesticides to aquatic microorganisms: A review. Environ Toxicol Chem 20(1):84–98. https://doi.org/10.1002/etc.5620200108
Domínguez-Álvarez E, Plano D, Font M et al (2014) Synthesis and antiproliferative activity of novel selenoester derivatives. Eur J Med Chem 73:153–166. https://doi.org/10.1016/j.ejmech.2013.11.034
Fráguas RM, Costa VA, Terra WC et al (2020) Toxicities of 4,5-dihydroisoxazoles against root-knot nematodes and in silico studies of their modes of action. J Agric Food Chem 68:523–529. https://doi.org/10.1021/acs.jafc.9b07839
Fujiwara S, Kambe N (2005) Thio-, seleno-, and telluro-carboxylic acid esters. In: Topics in current chemistry, pp 87–140. https://doi.org/10.1007/b101007
Giannakou IO, Panopoulou S (2019) The use of fluensulfone for the control of root-knot nematodes in greenhouse cultivated crops: efficacy and phytotoxicity effects. Cogent Food Agric 5:1643819. https://doi.org/10.1080/23311932.2019.1643819
Hajdú G, Gecse E, Taisz I et al (2021) Toxic stress-specific cytoprotective responses regulate learned behavioral decisions in C. elegans. BMC Biol 19:26. https://doi.org/10.1186/s12915-021-00956-y
He L, He T, Farrar S et al (2017) Antioxidants maintain cellular redox homeostasis by elimination of reactive oxygen species. Cell Physiol Biochem 44:532–553. https://doi.org/10.1159/000485089
Hussey RS, Barker KR (1973) A comparison of methods of collecting inocula of Meloidogyne spp., including a new technique. Plant Disease Rep 57(12):1025–1028
Ignasiak K, Maxwell A (2017) Galleria mellonella (greater wax moth) larvae as a model for antibiotic susceptibility testing and acute toxicity trials. BMC Res Notes 10:428. https://doi.org/10.1186/s13104-017-2757-8
Jia H, Guo W, Li W et al (2019) Design, synthesis, and nematicidal activities of novel 1,3-thiazin(thiazol)-4-one derivatives against Meloidogyne incognita. J Chem Res 43:161–169. https://doi.org/10.1177/1747519819857506
Jones JT, Haegeman A, Danchin EGJ et al (2013) Top 10 plant-parasitic nematodes in molecular plant pathology. Mol Plant Pathol 14:946–961. https://doi.org/10.1111/mpp.12057
Kearn J, Ludlow E, Dillon J et al (2014) Fluensulfone is a nematicide with a mode of action distinct from anticholinesterases and macrocyclic lactones. Pestic Biochem Physiol 109:44–57. https://doi.org/10.1016/j.pestbp.2014.01.004
Keshavarz MH, Shirazi Z, Eskandari P (2022) A simple assessment of toxicity towards Chlorella vulgaris of organic aromatic compounds in environmental protection. Process Saf Environ Prot 163:669–678. https://doi.org/10.1016/j.psep.2022.05.074
Kumari S, Singh R, Kumar A, Walia RK (2014) Synthesis and nematicidal bioevaluation of substituted 2H–1-benzopyrane-2-ones and their carbamate derivatives against root-knot nematode (Meloidogyne javanica). Asian J Chem 26:3139–3143. https://doi.org/10.14233/ajchem.2014.15840
Lenardão EJ, Santi C, Sancineto L (2018) New frontiers in organoselenium compounds. Springer, Cham. https://doi.org/10.1007/978-3-319-92405-2
Lu P, Davis RF, Kemerait RC, van Iersel MW, Scherm H et al (2014) Physiological effects of Meloidogyne incognita infection on cotton genotypes with differing levels of resistance in the greenhouse. J Nematol 46(4):352–359
Margie O, Palmer C, Chin-Sang I (2013) C. elegans chemotaxis assay. J Visual Exp. https://doi.org/10.3791/50069
Marin G, Braga AL, Rosa AS et al (2009) Efficient synthesis of selenol esters from acid chlorides mediated by indium metal. Tetrahedron 65:4614–4618. https://doi.org/10.1016/j.tet.2009.03.069
Mateille T, Tavoillot J, Goillon C et al (2020) Competitive interactions in plant-parasitic nematode communities affecting organic vegetable cropping systems. Crop Prot 135:105206. https://doi.org/10.1016/j.cropro.2020.105206
Maurya R, Dubey K, Singh D et al (2019) Effect of difenoconazole fungicide on physiological responses and ultrastructural modifications in model organism Tetrahymena pyriformis. Ecotoxicol Environ Saf 182:109375. https://doi.org/10.1016/j.ecoenv.2019.109375
Moens M, Perry RN, Starr JL (2009) Meloidogyne species—a diverse group of novel and important plant parasites. Root-knot nematodes. CABI, pp 1–17. https://doi.org/10.1079/9781845934927.0001
Neto MF, Nguyen QH, Marsili J et al (2016) The nematode Caenorhabditis elegans displays a chemotaxis behavior to tuberculosis-specific odorants. J Clin Tuberc Other Mycobact Dis 4:44–49. https://doi.org/10.1016/j.jctube.2016.06.001
Ništiar F, Rácz O, Brenišin M (2016) Can imprinting play a role in the response of Tetrahymena pyriformis to toxic substance exposure? Environ Epigenet 2:dvw010. https://doi.org/10.1093/eep/dvw010
Nitao JK, Meyer SL, Chitwood DJ (1999) In-vitro assays of Meloidogyne incognita and heterodera glycines for detection of nematode-antagonistic fungal compounds. J Nematol 31(2):172–183
Oka Y (2020) From old-generation to next-generation nematicides. Agronomy 10:1387. https://doi.org/10.3390/agronomy10091387
Oliveira AEMFM, Duarte JL, Cruz RAS et al (2017) Pterodon emarginatus oleoresin-based nanoemulsion as a promising tool for Culex quinquefasciatus (Diptera: Culicidae) control. J Nanobiotechnology 15:2. https://doi.org/10.1186/s12951-016-0234-5
Oliveira DF, Costa VA, Terra WC et al (2019) Impact of phenolic compounds on Meloidogyne incognita in vitro and in tomato plants. Exp Parasitol 199:17–23. https://doi.org/10.1016/j.exppara.2019.02.009
Pacheco AG, Rebelo MdeF (2013) A simple R-based function to estimate lethal concentrations. Mar Environ Res 91:41–44. https://doi.org/10.1016/j.marenvres.2013.08.003
Palomares-Rius JE, Escobar C, Cabrera J et al (2017) Anatomical alterations in plant tissues induced by plant-parasitic nematodes. Front Plant Sci. https://doi.org/10.3389/fpls.2017.01987
Pan L, Li X-Z, Sun D-A et al (2016) Design and synthesis of novel coumarin analogs and their nematicidal activity against five phytonematodes. Chin Chem Lett 27:375–379. https://doi.org/10.1016/j.cclet.2016.01.029
Papp D, Csermely P, Sőti C (2012) A Role for SKN-1/Nrf in pathogen resistance and immunosenescence in Caenorhabditis elegans. PLoS Pathog 8:e1002673. https://doi.org/10.1371/journal.ppat.1002673
Park M-G, Choi J, Hong Y-S et al (2020) Negative effect of methyl bromide fumigation work on the central nervous system. PLoS ONE 15:e0236694. https://doi.org/10.1371/journal.pone.0236694
Ploeg A, Stoddard S, Becker JO (2019) Control of Meloidogyne incognita in sweetpotato with fluensulfone. J Nematol 51:1–8. https://doi.org/10.21307/jofnem-2019-018
Porta-de-la-Riva M, Fontrodona L, Villanueva A, Cerón J (2012) Basic Caenorhabditis elegans methods: synchronization and observation. J vis Exp. https://doi.org/10.3791/4019
Quines CB, Rosa SG, Velasquez D et al (2018) (p-ClPhSe)2 stabilizes metabolic function in a rat model of neuroendocrine obesity induced by monosodium glutamate. Food Chem Toxicol 118:168–180. https://doi.org/10.1016/j.fct.2018.05.010
Ramarao N, Nielsen-Leroux C, Lereclus D (2012) The insect Galleria mellonella as a powerful infection model to investigate bacterial pathogenesis. J vis Exp. https://doi.org/10.3791/4392
Regmi H, Desaeger J (2020) Integrated management of root-knot nematode (Meloidogyne spp) in Florida tomatoes combining host resistance and nematicides. Crop Protect 134:105170. https://doi.org/10.1016/j.cropro.2020.105170
Reich HJ, Cohen ML, Clark PS (2003) Reagents for synthesis of organoselenium compounds: diphenyl diselenide and benzeneselenenyl chloride. In: Organic syntheses. Wiley, pp 141–141. https://doi.org/10.1002/0471264180.os059.20
Sanadhya P, Bucki P, Liarzi O et al (2018) Caenorhabditis elegans susceptibility to Daldinia cf. concentrica bioactive volatiles is coupled with expression activation of the stress-response transcription factor daf-16, a part of distinct nematicidal action. PLoS ONE 13:10196870. https://doi.org/10.1371/journal.pone.0196870
Sancineto L, Pinto Vargas J, Monti B et al (2017) Atom efficient preparation of zinc selenates for the synthesis of selenol esters under “On Water” conditions. Molecules 22:953. https://doi.org/10.3390/molecules22060953
Sanmartín C, Plano D, Domínguez E et al (2009) Synthesis and pharmacological screening of several aroyl and heteroaroyl selenylacetic acid derivatives as cytotoxic and antiproliferative agents. Molecules 14:3313–3338. https://doi.org/10.3390/molecules14093313
Schouteden N, Lemmens E, Stuer N et al (2017) Direct nematicidal effects of methyl jasmonate and acibenzolar-S-methyl against Meloidogyne incognita. Nat Prod Res 31:1219–1222. https://doi.org/10.1080/14786419.2016.1230111
Shukla N, Yadav R, Kaur P et al (2018) Transcriptome analysis of root-knot nematode (Meloidogyne incognita) -infected tomato (Solanum lycopersicum) roots reveals complex gene expression profiles and metabolic networks of both host and nematode during susceptible and resistance responses. Mol Plant Pathol 19:615–633. https://doi.org/10.1111/mpp.12547
Silva FJ, Campos VP, Oliveira DF et al (2019) Chalcone analogues: synthesis, activity against Meloidogyne incognita, and in silico interaction with cytochrome P450. J Phytopathol 167:197–208. https://doi.org/10.1111/jph.12787
Souza H, de Sousa R, Lira B et al (2018) Synthesis, in silico study and antimicrobial evaluation of new selenoglycolicamides. J Braz Chem Soc. https://doi.org/10.21577/0103-5053.20180148
Stefanello ST, Gubert P, Puntel B et al (2015) Protective effects of novel organic selenium compounds against oxidative stress in the nematode Caenorhabditis elegans. Toxicol Rep 2:961–967. https://doi.org/10.1016/j.toxrep.2015.06.010
Targanski SK, Sousa JR, Stein ALA, Soares MA (2021) Selective activity of diselenides against Aedes aegypti (Diptera: Culicidae) larvae. Rev Soc Bras Med Trop. https://doi.org/10.1590/0037-8682-0146-2020
Taylor A, Sasser J (1978) Biology, identification and control of root-knot nematodes (Meloidogyne species). Raleigh. NC: United States Agency for International
Temperini A, Piazzolla F, Minuti L et al (2017) General, mild, and metal-free synthesis of phenyl selenoesters from anhydrides and their use in peptide synthesis. J Org Chem 82:4588–4603. https://doi.org/10.1021/acs.joc.7b00173
Thomas RJ, Hamblin KA, Armstrong SJ et al (2013) Galleria mellonella as a model system to test the pharmacokinetics and efficacy of antibiotics against Burkholderia pseudomallei. Int J Antimicrob Agents 41:330–336. https://doi.org/10.1016/j.ijantimicag.2012.12.009
Vanegas JAG, Pacule HB, Capitão RM et al (2022) Methyl esters of (E)-cinnamic acid: activity against the plant-parasitic nematode Meloidogyne incognita and In Silico Interaction with Histone Deacetylase. J Agric Food Chem 70:6624–6633. https://doi.org/10.1021/acs.jafc.1c08142
Victoria FN, Martinez DM, Castro M et al (2013) Antioxidant properties of (R)-Se-aryl thiazolidine-4-carboselenoate. Chem Biol Interact 205:100–107. https://doi.org/10.1016/j.cbi.2013.06.019
Vos C, Schouteden N, van Tuinen D et al (2013) Mycorrhiza-induced resistance against the root–knot nematode Meloidogyne incognita involves priming of defense gene responses in tomato. Soil Biol Biochem 60:45–54. https://doi.org/10.1016/j.soilbio.2013.01.013
Waldo BD, Grabau ZJ, Mengistu TM, Crow WT (2019) Nematicide effects on non-target nematodes in bermudagrass. J Nematol 51:1–12. https://doi.org/10.21307/jofnem-2019-009
Whitehead AG, Hemming JR (1965) A comparison of some quantitative methods of extracting small vermiform nematodes from soil. Ann Appl Biol 55:25–38. https://doi.org/10.1111/j.1744-7348.1965.tb07864.x
WHO (2005) Guidelines for laboratory and field testing of mosquito larvicides. No. WHO/CDS/WHOPES/GCDPP/2005.13. World Health Organization
Wram CL, Zasada IA (2019) Short-term effects of sublethal doses of nematicides on Meloidogyne incognita. Phytopathology 109:1605–1613. https://doi.org/10.1094/PHYTO-11-18-0420-R
Yue X, Li F, Wang B (2020) Activity of four nematicides against Meloidogyne incognita race 2 on tomato plants. J Phytopathol 168:399–404. https://doi.org/10.1111/jph.12904
Zamberlan DC, Arantes LP, Machado ML et al (2014) Diphenyl-diselenide suppresses amyloid-β peptide in Caenorhabditis elegans model of Alzheimer’s disease. Neuroscience 278:40–50. https://doi.org/10.1016/j.neuroscience.2014.07.068
Zborowski VA, Sari MHM, Heck SO et al (2016) p-Chloro-diphenyl diselenide reverses memory impairment-related to stress caused by corticosterone and modulates hippocampal [3H]glutamate uptake in mice. Physiol Behav 164:25–33. https://doi.org/10.1016/j.physbeh.2016.05.029
Acknowledgements
We thank the researchers Dr Margareth de Lara Capurro Guimarães from the Institute of Biomedical Sciences at the University of São Paulo (ICB – USP, São Paulo, SP, Brazil) for providing Ae. aegypti eggs; Dr. Adriano Nunes Nesi, from the Department of Plant Biology at the Federal University of Viçosa (UFV, Viçosa, MG, Brazil) for supplying the microalgae; Dra. Marcia Cristina de Azevedo Prata from the Brazilian Agricultural Research Corporation (Embrapa, Brasília, DF, Brazil) for supplying G. mellonella eggs; and Dr. Tiago Campos Pereira from the Department of Biology at the University of São Paulo (FFCLRP—USP, Ribeirão Preto, SP, Brazil) for providing the C. elegans N2 strain. We thank our laboratory colleagues, in particular Jaqueline Alves Senabio, MD, for her availability to read and revise important parts of this manuscript.
Funding
This project was funded by a scholarship from the National Council for Scientific and Technological Development (CNPq, Grant #133463/2019-2), as well as research support grants from CNPq (Grant # 426986/2018-0) and the Research Support Foundation from Mato Grosso State (FAPEMAT, Grant # 0583867/2016).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Communicated by Murray Isman.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Vendruscolo, S.J., de Oliveira, A.J., de Sousa, J.R. et al. Nematicidal and ovicidal activity of environmentally-friendly selenol ester derivatives against Meloidogyne incognita. J Pest Sci (2024). https://doi.org/10.1007/s10340-023-01732-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10340-023-01732-3