Abstract
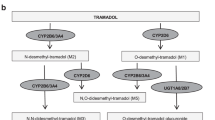
Codeine is metabolized by the CYP2D6 enzyme, and individuals with certain genetic variations of the CYP2D6 gene may metabolize codeine differently, leading to variable efficacy and toxicity. Drug-drug interactions can also affect the metabolism of codeine. A tool to adjust codeine dose based on these factors does not currently exist. Healthcare providers should use their clinical judgment and reference different established dosing guidelines to determine the appropriate dose of codeine for individual patients. The study provides a tool that assists prescribers in adjusting codeine dose based on CYP2D6 gene-pair polymorphisms and drug-drug interactions. Highlighted is the need to consider pharmacogenetics and drug-drug interactions when determining the appropriate dosing of codeine and provide a framework for implementing individualized dosing based on these factors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 print issues and online access
$259.00 per year
only $43.17 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
How codeine metabolism affects its clinical use. Pharm J [Internet]. 2021 [cited 2023 Jul 25]. Available from: https://pharmaceutical-journal.com/article/ld/how-codeine-metabolism-affects-its-clinical-use.
Lam J, Woodall KL, Solbeck P, Ross CJD, Carleton BC, Hayden MR, et al. Codeine-related deaths: the role of pharmacogenetics and drug interactions. Forensic Sci Int. 2014;239:50–6.
Crews KR, Gaedigk A, Dunnenberger HM, Leeder JS, Klein TE, Caudle KE, et al. Clinical pharmacogenetics implementation consortium guidelines for cytochrome P450 2D6 genotype and codeine therapy: 2014 update. Clin Pharm Ther. 2014;95:376–82.
Chidambaran V, Sadhasivam S, Mahmoud M. Codeine and opioid metabolism: implications and alternatives for pediatric pain management. Curr Opin Anaesthesiol. 2017;30:349–56.
Codeine label. Last accessed December 2022 [Internet]. [cited 2023 Apr 4]. Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/022402s009lbl.pdf.
Adam de Beaumais T, Jacqz-Aigrain E. Pharmacogenetics: applications to pediatric patients. In: Advances in pharmacology [Internet]. Elsevier; 2018. p. 191–215. Available from: https://linkinghub.elsevier.com/retrieve/pii/S1054358918300255.
Ma TKW, Lam YY, Tan VP, Yan BP. Variability in response to clopidogrel: how important are pharmacogenetics and drug interactions?: Clopidogrel response variability. Br J Clin Pharm. 2011;72:697–706.
Saab Y, Zeenny R, Ramadan W. Optimizing clopidogrel dose response: a new clinical algorithm comprising CYP2C19 pharmacogenetics and drug interactions. Ther Clin Risk Manag. 2015;11:1421–7.
Fulton CR, Zang Y, Desta Z, Rosenman MB, Holmes AM, Decker BS, et al. Drug–gene and drug–drug interactions associated with tramadol and codeine therapy in the INGENIOUS trial. Pharmacogenomics. 2019;20:397–408.
FDA table of substrates. Last accessed December 2022 [Internet]. [cited 2023 Apr 4]. Available from: https://www.fda.gov/drugs/drug-interactions-labeling/drug-development-and-drug-interactions-table-substrates-inhibitors-and-inducers.
Lunenburg CATC, van der Wouden CH, Nijenhuis M, Crommentuijn-van Rhenen MH, de Boer-Veger NJ, Buunk AM, et al. Dutch Pharmacogenetics Working Group (DPWG) guideline for the gene–drug interaction of DPYD and fluoropyrimidines. Eur J Hum Genet. 2020;28:508–17.
Ross CJD, Visscher H, Sistonen J, Brunham LR, Pussegoda K, Loo TT, et al. The Canadian Pharmacogenomics Network for Drug Safety: a model for safety pharmacology. Thyroid. 2010;20:681–7.
Abdullah-Koolmees H, van Keulen AM, Nijenhuis M, Deneer VHM. Pharmacogenetics guidelines: overview and comparison of the DPWG, CPIC, CPNDS, and RNPGx guidelines. Front Pharm. 2021;11:595219.
Whirl‐Carrillo M, Huddart R, Gong L, Sangkuhl K, Thorn CF, Whaley R, et al. An evidence‐based framework for evaluating pharmacogenomics knowledge for personalized medicine. Clin Pharm Ther. 2021;110:563–72.
Author information
Authors and Affiliations
Contributions
Both authors contributed equally to the conceptualization and design of the study, modeling of response, critical analysis, and write up.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Saab, Y., Nakad, Z. A software tool to adjust codeine dose based on CYP2D6 gene-pair polymorphisms and drug-drug interactions. Pharmacogenomics J 23, 217–224 (2023). https://doi.org/10.1038/s41397-023-00318-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41397-023-00318-7