Bioorganic Chemistry ( IF 5.1 ) Pub Date : 2018-09-08 , DOI: 10.1016/j.bioorg.2018.09.015 Sana Tariq , Payal Kamboj , Ozair Alam , Mohd. Amir
|
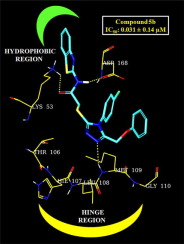
Novel N-(benzothiazol/oxazol-2-yl)-2-[(5-(phenoxymethyl)-4-aryl-4H-1,2,4-triazol-3-yl)thio] acetamide derivatives (5a-n) were synthesized and investigated for in vitro anti-inflammatory activity and p38α MAP kinase inhibition. Compounds showing good in vitro activities (5a, 5b, 5d, 5e, 5i, 5k and 5l) were studied for their in vivo anti-inflammatory activity using carrageenan induced rat paw edema model. Compound 5b emerged as the most active compound with an edema inhibition of 84.43%. It also showed improved GI safety profile with lower ulcer severity index and lipid peroxidation potential. Also, p38α MAP kinase assay of 5b showed superior inhibitory potency (IC50:0.031 ± 0.14 µM) than the standard SB 203580 (IC50:0.043 ± 0.14 µM). To predict their binding mode compounds were also docked against p38α MAP kinase enzyme. Compound 5b and SB 203580 showed hinge region interaction with MET 109.
中文翻译:

1,2,4-三唑基苯并噻唑/苯并恶唑衍生物:设计,合成,p38αMAP激酶抑制,抗炎活性和分子对接研究
新的N-(苯并噻唑/恶唑-2-基)-2-[(5-(苯氧基甲基)-4-芳基-4H-1,2,4-三唑-3-基)硫代]乙酰胺衍生物(5a-n)合成并研究其体外抗炎活性和p38αMAP激酶抑制作用。使用角叉菜胶诱导的大鼠爪水肿模型研究了显示出良好体外活性(5a,5b,5d,5e,5i,5k和5l)的化合物的体内抗炎活性。化合物5b出现为最活跃的化合物,具有84.43%的水肿抑制作用。它还显示出改善的胃肠道安全性,具有较低的溃疡严重程度指数和脂质过氧化潜力。同样,5b的p38αMAP激酶测定显示出比标准SB 203580(IC 50:0.043±0.14 µM)优越的抑制力(IC 50:0.031 ±0.14 µM)。为了预测它们的结合模式,化合物也与p38αMAP激酶对接。化合物5b和SB 203580显示出与MET 109的铰链区相互作用。



























 京公网安备 11010802027423号
京公网安备 11010802027423号