Tetrahedron Letters ( IF 1.8 ) Pub Date : 2018-06-20 , DOI: 10.1016/j.tetlet.2018.06.047 Hao-Fei Yu , Wan-Yi Huang , Cai-Feng Ding , Xin Wei , Lan-Chun Zhang , Xu-Jie Qin , Hong-Xia Ma , Zi-Feng Yang , Ya-Ping Liu , Rong-Ping Zhang , Xin-Hua Wang , Xiao-Dong Luo
|
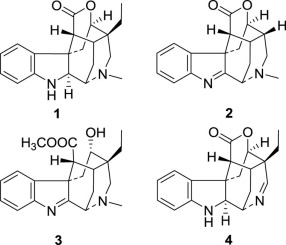
Three new cage-like monoterpenoid indole alkaloids, scholarisines T–V (1–3), together with three known analogues 4–6 were isolated from the leaves of Alstonia scholaris. Among them, 2 represents a unique degraded derivative, whereas 3 shares a rare 5,16-seco lactone scaffold. The structures were mainly established by extensive spectroscopic data analyses, and their plausible biosynthesis pathway from picrinine were proposed. Compared with positive control cefotaxime, alkaloid 2 showed remarkable antibacterial activity against Bacillus subtilis with an MIC value of 3.12 μg/mL, whereas 1–3 exhibited significant antibacterial effects on Escherichia coli with an MIC value of 0.78 μg/mL.
中文翻译:

笼状与抗菌活性的单萜吲哚生物碱灯台
三个新的笼状单萜吲哚生物碱,scholarisines T-V(1 - 3),具有三个已知类似物一起4 - 6是从叶分离的灯台树。其中,2代表独特的降解衍生物,而3代表稀有的5,16- seco内酯支架。该结构主要是通过广泛的光谱数据分析来建立的,并提出了它们从苦味碱的合理生物合成途径。与阳性对照头孢噻肟相比,生物碱2对枯草芽孢杆菌具有显着的抗菌活性与3.12微克/毫升的MIC值,而1 - 3显示对显著抗菌作用大肠杆菌具有0.78的MIC值微克/毫升。


























 京公网安备 11010802027423号
京公网安备 11010802027423号