Chem ( IF 23.5 ) Pub Date : 2018-06-07 , DOI: 10.1016/j.chempr.2018.05.007 Hugo Rego Campello , Silvia G. Del Villar , Aurélien Honraedt , Teresa Minguez , A. Sofia F. Oliveira , Kara E. Ranaghan , Deborah K. Shoemark , Isabel Bermudez , Cecilia Gotti , Richard B. Sessions , Adrian J. Mulholland , Susan Wonnacott , Timothy Gallagher
|
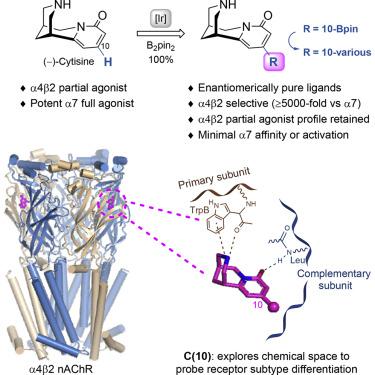
Differentiating nicotinic acetylcholine receptors (nAChR) to target the high-affinity nicotine α4β2 subtype is a major challenge in developing effective addiction therapies. Although cytisine 1 and varenicline 2 (current smoking-cessation agents) are partial agonists of α4β2, these drugs display full agonism at the α7 nAChR subtype. Site-specific modification of (−)-cytisine via Ir-catalyzed C‒H activation provides access to C(10) variants 6–10, 13, 14, 17, 20, and 22, and docking studies reveal that C(10) substitution targets the complementary region of the receptor binding site, mediating subtype differentiation. C(10)-modified cytisine ligands retain affinity for α4β2 nAChR and are partial agonists, show enhanced selectivity for α4β2 versus both α3β4 and α7 subtypes, and critically, display negligible activity at α7. Molecular dynamics simulations link the C(10) moiety to receptor subtype differentiation; key residues beyond the immediate binding site are identified, and molecular-level conformational behavior responsible for these crucial differences is characterized.
中文翻译:

通过(-)-Cytisine的直接CizationH功能化释放烟碱选择性
区分烟碱型乙酰胆碱受体(nAChR)以靶向高亲和力尼古丁α4β2亚型是开发有效成瘾疗法的主要挑战。尽管胱氨酸1和伐尼克兰2(目前的戒烟药物)是α4β2的部分激动剂,但这些药物对α7nAChR亚型表现出完全的激动作用。( - ) -的位点特异性修饰经由铱催化的C-H活化金雀花碱提供了访问C(10)变体6 - 10,13,14,17,20,和22,和对接研究表明C(10)替代靶向受体结合位点的互补区域,介导亚型分化。C(10)修饰的胱氨酸配体保留对α4β2nAChR的亲和力,并且是部分激动剂,与α3β4和α7亚型相比,对α4β2的选择性增强,并且关键地,对α7的活性微不足道。分子动力学模拟将C(10)部分链接到受体亚型分化。确定了超出直接结合位点的关键残基,并表征了导致这些关键差异的分子水平构象行为。


























 京公网安备 11010802027423号
京公网安备 11010802027423号