Bioorganic & Medicinal Chemistry Letters ( IF 2.7 ) Pub Date : 2018-05-26 , DOI: 10.1016/j.bmcl.2018.05.050 Fei Li , Xi-Xi Guo , Guang-Zhi Zeng , Wei-Wei Qin , Bo Zhang , Ning-Hua Tan
|
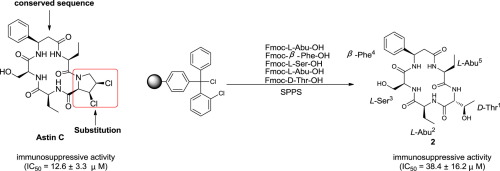
To further investigate on the structure-activity relationships of immunosuppressive Astin C, seventeen analogues 1–17 were designed and synthetized via amino acid substitution strategy by the solid-phase peptide synthesis method for the first time. In comparison with Astin C (IC50 = 12.6 ± 3.3 μM), only compounds 2 (IC50 = 38.4 ± 16.2 μM), 4 (IC50 = 51.8 ± 12.7 μM), 5 (IC50 = 65.2 ± 15.6 μM), and 8 (IC50 = 61.8 ± 12.4 μM) exhibited immunosuppressive activity in the Lymph node cells of mice. These results showed that the Astin C analogues containing D-amino acid residues, hydrophobic long-chain alkyl substituents, and aryl substituents performed better than those carrying hydrophilic amino acid residues and short-chain alkyl substituents. Moreover compounds 15, 16, and 17 had no immunosuppressive activity, which suggested that cis-3,4-dichlorinated proline played an important role in the immunosuppressive activity of Astin C.
中文翻译:

植物环肽Astin C类似物的设计与合成及其免疫抑制活性的研究
对免疫抑制阿斯廷C的结构-活性关系进一步调查,17个类似物1 - 17设计并synthetized经由通过首次固相肽合成方法的氨基酸取代的策略。与Astin C(IC 50 = 12.6±3.3μM)相比,仅化合物2(IC 50 = 38.4±16.2μM),4(IC 50 = 51.8±12.7μM),5(IC 50 = 65.2±15.6μM),和8(IC 50 = 61.8±12.4μM)在小鼠的淋巴结细胞中表现出免疫抑制活性。这些结果表明,含有D-氨基酸残基,疏水性长链烷基取代基和芳基取代基的Astin C类似物的性能优于带有亲水性氨基酸残基和短链烷基取代基的那些。此外化合物15,16和17没有免疫抑制活性,这表明顺式-3,4-二氯代脯氨酸阿斯廷C的免疫抑制活性中起重要作用



























 京公网安备 11010802027423号
京公网安备 11010802027423号