当前位置:
X-MOL 学术
›
RSC Med. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Evaluation of A-ring fused pyridine d-modified androstane derivatives for antiproliferative and aldo–keto reductase 1C3 inhibitory activity†
RSC Medicinal Chemistry ( IF 4.1 ) Pub Date : 2018-04-30 00:00:00 , DOI: 10.1039/c8md00077h Marina P Savić 1 , Jovana J Ajduković 1 , Jovana J Plavša 2 , Sofija S Bekić 1 , Andjelka S Ćelić 2 , Olivera R Klisurić 3 , Dimitar S Jakimov 4 , Edward T Petri 2 , Evgenija A Djurendić 1
RSC Medicinal Chemistry ( IF 4.1 ) Pub Date : 2018-04-30 00:00:00 , DOI: 10.1039/c8md00077h Marina P Savić 1 , Jovana J Ajduković 1 , Jovana J Plavša 2 , Sofija S Bekić 1 , Andjelka S Ćelić 2 , Olivera R Klisurić 3 , Dimitar S Jakimov 4 , Edward T Petri 2 , Evgenija A Djurendić 1
Affiliation
|
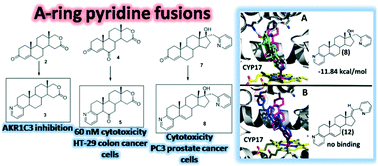
New A-ring pyridine fused androstanes in 17a-homo-17-oxa (D-homo lactone), 17α-picolyl or 17(E)-picolinylidene series were synthesized and validated by X-ray crystallography, HRMS, IR and NMR spectroscopy. Novel compounds 3, 5, 8 and 12 were prepared by treatment of 4-en-3-one or 4-ene-3,6-dione D-modified androstane derivatives with propargylamine catalyzed by Cu(II), and evaluated for potential anticancer activity in vitro using human cancer cell lines and recombinant targets of steroidal anti-cancer drugs. Pyridine fusion to position 3,4 of the A-ring may dramatically enhance affinity of 17α-picolyl compounds for CYP17 while conferring selective antiproliferative activity against PC-3 cells. Similarly, pyridine fusion to the A-ring of steroidal D-homo lactones led to identification of new inhibitors of aldo–keto reductase 1C3, an enzyme targeted in acute myeloid leukemia, breast and prostate cancers. One A-pyridine D-lactone steroid 5 also has selective submicromolar antiproliferative activity against HT-29 colon cancer cells. None of the new derivatives have affinity for estrogen or androgen receptors in a yeast screen, suggesting negligible estrogenicity and androgenicity. Combined, our results suggest that A-ring pyridine fusions have potential in modulating the anticancer activity of steroidal compounds.
中文翻译:

评估 A 环稠合吡啶 d-修饰的雄甾烷衍生物的抗增殖和醛酮还原酶 1C3 抑制活性†
合成了 17a-homo-17-oxa ( D -homo lactone)、17α-甲基吡啶或 17( E )-甲基吡啶系列中的新型 A 环吡啶稠合雄甾烷,并通过 X 射线晶体学、HRMS、IR 和 NMR 光谱学进行了验证。新型化合物3 , 5 , 8和12通过 Cu( II )催化的炔丙基胺处理 4-en-3-one 或 4-ene-3,6-dione D-修饰的雄甾烷衍生物制备,并评估其潜在的抗癌作用体外活性使用人类癌细胞系和甾体抗癌药物的重组靶点。吡啶与 A 环的 3,4 位融合可显着增强 17α-吡啶甲基化合物对 CYP17 的亲和力,同时赋予对 PC-3 细胞的选择性抗增殖活性。类似地,吡啶与甾体D-同型内酯的 A 环融合导致发现了醛酮还原酶 1C3 的新抑制剂,这是一种针对急性髓性白血病、乳腺癌和前列腺癌的酶。一A-吡啶D-内酯类固醇5对 HT-29 结肠癌细胞还具有选择性的亚微摩尔抗增殖活性。在酵母筛选中,没有一种新衍生物对雌激素或雄激素受体具有亲和力,这表明雌激素和雄激素可以忽略不计。综合起来,我们的结果表明,A 环吡啶融合体在调节甾体化合物的抗癌活性方面具有潜力。
更新日期:2018-04-30
中文翻译:

评估 A 环稠合吡啶 d-修饰的雄甾烷衍生物的抗增殖和醛酮还原酶 1C3 抑制活性†
合成了 17a-homo-17-oxa ( D -homo lactone)、17α-甲基吡啶或 17( E )-甲基吡啶系列中的新型 A 环吡啶稠合雄甾烷,并通过 X 射线晶体学、HRMS、IR 和 NMR 光谱学进行了验证。新型化合物3 , 5 , 8和12通过 Cu( II )催化的炔丙基胺处理 4-en-3-one 或 4-ene-3,6-dione D-修饰的雄甾烷衍生物制备,并评估其潜在的抗癌作用体外活性使用人类癌细胞系和甾体抗癌药物的重组靶点。吡啶与 A 环的 3,4 位融合可显着增强 17α-吡啶甲基化合物对 CYP17 的亲和力,同时赋予对 PC-3 细胞的选择性抗增殖活性。类似地,吡啶与甾体D-同型内酯的 A 环融合导致发现了醛酮还原酶 1C3 的新抑制剂,这是一种针对急性髓性白血病、乳腺癌和前列腺癌的酶。一A-吡啶D-内酯类固醇5对 HT-29 结肠癌细胞还具有选择性的亚微摩尔抗增殖活性。在酵母筛选中,没有一种新衍生物对雌激素或雄激素受体具有亲和力,这表明雌激素和雄激素可以忽略不计。综合起来,我们的结果表明,A 环吡啶融合体在调节甾体化合物的抗癌活性方面具有潜力。

























 京公网安备 11010802027423号
京公网安备 11010802027423号