当前位置:
X-MOL 学术
›
Catal. Sci. Technol.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Halogen type as a selectivity switch in catalysed alkane oxyhalogenation†
Catalysis Science & Technology ( IF 5 ) Pub Date : 2018-04-03 00:00:00 , DOI: 10.1039/c8cy00122g Guido Zichittella 1, 2, 3, 4, 5 , Begoña Puértolas 1, 2, 3, 4, 5 , Vladimir Paunović 1, 2, 3, 4, 5 , Theresa Block 6, 7, 8, 9 , Rainer Pöttgen 6, 7, 8, 9 , Javier Pérez-Ramírez 1, 2, 3, 4, 5
Catalysis Science & Technology ( IF 5 ) Pub Date : 2018-04-03 00:00:00 , DOI: 10.1039/c8cy00122g Guido Zichittella 1, 2, 3, 4, 5 , Begoña Puértolas 1, 2, 3, 4, 5 , Vladimir Paunović 1, 2, 3, 4, 5 , Theresa Block 6, 7, 8, 9 , Rainer Pöttgen 6, 7, 8, 9 , Javier Pérez-Ramírez 1, 2, 3, 4, 5
Affiliation
|
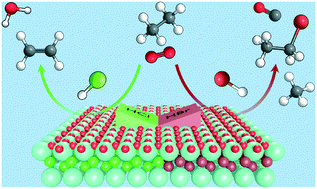
Catalysed alkane oxyhalogenation has recently been demonstrated as an attractive route for the selective functionalisation of natural gas into commodity chemicals such as alkyl halides or olefins. Herein, we assess the role of the hydrogen halide on the product distribution in ethane and propane oxyhalogenation over europium oxychloride and europium oxybromide catalysts. While olefins are the main reaction products in oxychlorination (selectivity ≥95%), the formation of alkyl bromides (selectivity ≤95%) is favoured in oxybromination, particularly at low conversion levels. At high conversion levels, olefins are also observed, although the selectivity is generally limited (≤60%) due to the formation of cracking and combustion products. A comprehensive kinetic analysis of the oxyhalogenation, halogenation, and oxidative dehydrogenation of the alkanes as well as of the dehydrohalogenation of the alkyl halides provide insights on the complex reaction network, which proceeds through alkyl halide formation followed by its conversion to olefins and/or halocarbons. The observed selectivity differences primarily originate from (i) the halide-dependent alkane activation path, which is surface-driven in oxychlorination, while it mostly occurs via gas-phase halogenation in oxybromination, along with (ii) the preferred tendency of alkyl chlorides towards catalytic dehydrochlorination pathways opposing alkyl bromides that undergo further bromination routes in the gas phase. Finally, detailed characterisation corroborated the above findings and revealed the higher structural stability of europium oxychloride compared to the analogous oxybromide phase under oxyhalogenation conditions. These results deepen the understanding of the alkane oxyhalogenation chemistry on catalyst surfaces for the manufacture of valuable commodities.
中文翻译:

卤素类型作为催化链烷氧基卤化反应中的选择性开关†
最近已证明,催化烷氧基卤代反应是将天然气选择性官能化为商品化学品(如烷基卤化物或烯烃)的一种有吸引力的途径。在本文中,我们评估了卤化氢在乙烷和丙烷氧卤化反应上在氯氧化和溴化氧化催化剂上产物分布中的作用。烯烃是氧氯化反应中的主要反应产物(选择性≥95%),而烷基溴化物的形成(选择性≤95%)在氧溴化反应中是有利的,特别是在低转化率的情况下。在高转化率下,虽然由于裂化和燃烧产物的形成,选择性通常受到限制(≤60%),但仍观察到烯烃。氧卤化,卤化,烷烃的氧化脱氢以及卤代烷的脱卤代氢提供了复杂的反应网络的见解,该网络通过卤代烷的形成随后转化为烯烃和/或卤代烃而得以进行。观察到的选择性差异主要来自(i)卤化物依赖性烷烃活化路径,该路径在氧氯化反应中是表面驱动的,而大多数情况下是这样发生的通过氧溴化中的气相卤化,以及(ii)烷基氯朝向与在气相中经历进一步溴化途径的烷基溴相反的催化脱氯化氢途径的优选趋势。最后,详细的表征证实了以上发现,并揭示了在卤代氧条件下,三氯氧化euro相较于类似的溴化氧相具有更高的结构稳定性。这些结果加深了对用于制造有价值的商品的催化剂表面上的烷氧基卤代化学的理解。
更新日期:2018-04-03
中文翻译:

卤素类型作为催化链烷氧基卤化反应中的选择性开关†
最近已证明,催化烷氧基卤代反应是将天然气选择性官能化为商品化学品(如烷基卤化物或烯烃)的一种有吸引力的途径。在本文中,我们评估了卤化氢在乙烷和丙烷氧卤化反应上在氯氧化和溴化氧化催化剂上产物分布中的作用。烯烃是氧氯化反应中的主要反应产物(选择性≥95%),而烷基溴化物的形成(选择性≤95%)在氧溴化反应中是有利的,特别是在低转化率的情况下。在高转化率下,虽然由于裂化和燃烧产物的形成,选择性通常受到限制(≤60%),但仍观察到烯烃。氧卤化,卤化,烷烃的氧化脱氢以及卤代烷的脱卤代氢提供了复杂的反应网络的见解,该网络通过卤代烷的形成随后转化为烯烃和/或卤代烃而得以进行。观察到的选择性差异主要来自(i)卤化物依赖性烷烃活化路径,该路径在氧氯化反应中是表面驱动的,而大多数情况下是这样发生的通过氧溴化中的气相卤化,以及(ii)烷基氯朝向与在气相中经历进一步溴化途径的烷基溴相反的催化脱氯化氢途径的优选趋势。最后,详细的表征证实了以上发现,并揭示了在卤代氧条件下,三氯氧化euro相较于类似的溴化氧相具有更高的结构稳定性。这些结果加深了对用于制造有价值的商品的催化剂表面上的烷氧基卤代化学的理解。

























 京公网安备 11010802027423号
京公网安备 11010802027423号