Journal of Inorganic Biochemistry ( IF 3.9 ) Pub Date : 2018-03-09 , DOI: 10.1016/j.jinorgbio.2018.03.004 Ngo Fung Lee , Dharmesh Patel , Haiyan Liu , Rui Zhang
|
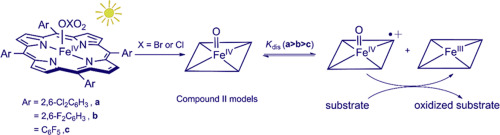
Iron(IV)-oxo porphyrins [FeIV(Por)O] (Por = poprhyrin), commonly called compound II models, were produced in three electron-deficient ligands by visible light irradiation of highly photo-labile porphyrin-iron(III) bromates or chlorates. The kinetics of oxygen transfer atom (OAT) reactions with aryl sulfides by these photo-generated [FeIV(Por)O] (3) were studied in CH3CN solutions. The iron(IV)-oxo porphyrins under study include 5,10,15,20-tetra(2,6-dichlorophenyl)porphyrin-iron(IV)-oxo (3a), 5,10,15,20-tetra(2,6-difluorophenyl)porphyrin-iron(IV)-oxo (3b), and 5,10,15,20-tetra(pentafluorophenyl)porphyrin-iron(IV)-oxo (3c). As expected, complexes 3 were competent oxidants and reacted rapidly with thioanisoles to give the corresponding sulfoxides with minor over-oxidation sulfones. Apparent second-order rate constants determined under pseudo-first-order conditions for sulfide oxidation reactions are (9.8 ± 0.1) × 102–(3.7 ± 0.3) × 101 M−1 s−1, which are 3 to 4 orders of magnitude greater in comparison to those of alkene epoxidations and activated CH bond oxidations by the same oxo species. Conventional Hammett analyses gave non-linear correlations, indicating no significant charge developed at the sulfur during the oxidation process. For a given substrate, the reactivity order for the iron(IV)-oxo species was 3c < 3b < 3a, which is inverted from expectations on the basis of the electron-withdrawing capacity of the porphyrin macrocycles. The absolute rate constants from kinetic studies provided insights into the transient oxidants in catalytic reactions under turnover conditions where actual reactive intermediates are not observable. Our kinetic and catalytic competition results strongly suggest that 3 may undergo a disproportionation reaction to form a higher oxidized iron(IV)-oxo porphyrin radical cations as the true oxidant.
中文翻译:

从光生化合物II模型的动力学研究中得出的见解:对芳基硫化物的反应性
铁(IV)-氧代卟啉[Fe IV(Por)O](Por = Poprhyrin),通常称为化合物II模型,是通过高度光不稳定的卟啉-铁(III)的可见光照射在三个缺电子配体中产生的溴酸盐或氯酸盐。在CH 3 CN溶液中研究了这些光生[Fe IV(Por)O](3)与芳基硫与氧转移原子(OAT)反应的动力学。研究中的铁(IV)-氧卟啉包括5,10,15,20-四(2,6-二氯苯基)卟啉-铁(IV)-氧(3a),5,10,15,20-四(2) 1,6-二氟苯基)卟啉-铁(IV)-氧(3b)和5,10,15,20-四(五氟苯基)卟啉-铁(IV)-氧(3c)。不出所料3种有效的氧化剂,可与硫代苯甲醚快速反应,得到相应的亚砜和少量的过氧化砜。在拟一级反应条件下,硫化物氧化反应确定的表观二级速率常数为(9.8±0.1)×10 2 –(3.7±0.3)×10 1 M -1 s -1,为3至4阶。与由相同的羰基化合物进行的烯烃环氧化和活化的C H键氧化相比,其抗氧化强度要高得多。传统的Hammett分析给出了非线性相关性,表明在氧化过程中在硫上没有产生明显的电荷。对于给定的底物,铁(IV)-氧代物种的反应顺序为3c < 3b < 3a,这是基于卟啉大环的吸电子能力而与预期相反的。动力学研究的绝对速率常数提供了在周转条件下无法观察到实际反应性中间体的催化反应中的瞬态氧化剂的见解。我们的动力学和催化竞争结果强烈表明3可能发生歧化反应以形成更高的氧化铁(IV)-氧代卟啉自由基阳离子作为真正的氧化剂。

























 京公网安备 11010802027423号
京公网安备 11010802027423号