当前位置:
X-MOL 学术
›
ACS Catal.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Kinetic Coupling of Water Splitting and Photoreforming on SrTiO3-Based Photocatalysts
ACS Catalysis ( IF 12.9 ) Pub Date : 2018-02-26 00:00:00 , DOI: 10.1021/acscatal.7b03192 Kai E. Sanwald 1 , Tobias F. Berto 1 , Andreas Jentys 1 , Donald M. Camaioni 2 , Oliver Y. Gutiérrez 2 , Johannes A. Lercher 1, 2
ACS Catalysis ( IF 12.9 ) Pub Date : 2018-02-26 00:00:00 , DOI: 10.1021/acscatal.7b03192 Kai E. Sanwald 1 , Tobias F. Berto 1 , Andreas Jentys 1 , Donald M. Camaioni 2 , Oliver Y. Gutiérrez 2 , Johannes A. Lercher 1, 2
Affiliation
|
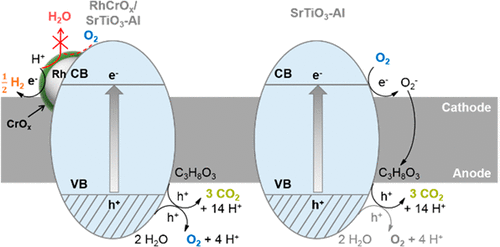
Coupling the proton reduction of overall water splitting with oxidation of oxygenated hydrocarbons (photoreforming) on Al-doped SrTiO3 decorated with cocatalysts enables efficient photocatalytic H2 generation along with oxygenate conversion, while decreasing the accumulation of harmful byproducts such as formaldehyde. Net H2 evolution rates result from the contributions of the individual rates of water oxidation, oxygenate oxidation, and the back-reaction of H2 and O2 to water. The latter reaction is suppressed by a RhCrOx cocatalyst or by high concentrations of oxygenates in the case of Rh cocatalyst, whereas the rates of organic oxidation depend on their molecular structure. In the absence of the back-reaction to water, the H2 evolution rates are independent of the oxygenate type and concentration because the rates of water splitting compensate the variations in the rates of oxygenate conversion. Under such conditions of suppressed back-reaction, the selectivities to water and oxygenate oxidation, both occurring with the same quantum efficiencies, depend on the oxygenate type and concentration. The dominant pathways for organic transformations are ascribed to the action of intermediates generated at the semiconductor during water oxidation and O2 evolution. On a semiconductor without cocatalyst, the O2 produced during overall water splitting is reductively activated to participate in oxidation of organics without consuming evolved H2.
中文翻译:

SrTiO 3基光催化剂上水分解和光重整的动力学耦合
在用助催化剂修饰的掺Al的SrTiO 3上,将总水分解的质子还原与含氧烃的氧化(光重整)相结合,可以有效地产生光催化的H 2以及含氧化合物,同时减少有害副产物(如甲醛)的积累。净H 2析出速率是由水氧化,氧合氧化和H 2和O 2与水的背反应的各个速率的贡献所决定的。RhCrO x抑制了后者的反应助催化剂或高浓度含氧化合物(在Rh助催化剂的情况下),而有机氧化的速率取决于其分子结构。在不存在对水的逆反应的情况下,H 2的析出速率与含氧化合物的类型和浓度无关,因为水的分解速率补偿了含氧化合物转化速率的变化。在这种抑制的后反应条件下,以相同的量子效率发生的对水和含氧化合物氧化的选择性取决于含氧化合物的类型和浓度。有机转化的主要途径归因于水氧化和O 2期间半导体中生成的中间体的作用进化。在没有助催化剂的半导体上,整个水分解过程中产生的O 2会被还原活化,以参与有机物的氧化,而不会消耗生成的H 2。
更新日期:2018-02-26
中文翻译:

SrTiO 3基光催化剂上水分解和光重整的动力学耦合
在用助催化剂修饰的掺Al的SrTiO 3上,将总水分解的质子还原与含氧烃的氧化(光重整)相结合,可以有效地产生光催化的H 2以及含氧化合物,同时减少有害副产物(如甲醛)的积累。净H 2析出速率是由水氧化,氧合氧化和H 2和O 2与水的背反应的各个速率的贡献所决定的。RhCrO x抑制了后者的反应助催化剂或高浓度含氧化合物(在Rh助催化剂的情况下),而有机氧化的速率取决于其分子结构。在不存在对水的逆反应的情况下,H 2的析出速率与含氧化合物的类型和浓度无关,因为水的分解速率补偿了含氧化合物转化速率的变化。在这种抑制的后反应条件下,以相同的量子效率发生的对水和含氧化合物氧化的选择性取决于含氧化合物的类型和浓度。有机转化的主要途径归因于水氧化和O 2期间半导体中生成的中间体的作用进化。在没有助催化剂的半导体上,整个水分解过程中产生的O 2会被还原活化,以参与有机物的氧化,而不会消耗生成的H 2。

























 京公网安备 11010802027423号
京公网安备 11010802027423号