Molecular Catalysis ( IF 4.6 ) Pub Date : 2018-02-21 , DOI: 10.1016/j.mcat.2018.01.031 Seunghee Pyen , Eunpyo Hong , Mi Shin , Young-Woong Suh , Chae-Ho Shin
|
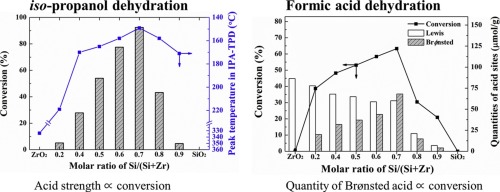
SiO2-ZrO2 is an interesting class of binary mixed oxide systems, but its acidity has not been intensively linked to the catalytic activity in the dehydration reactions. Thus, SiO2-ZrO2 mixed oxides with the Si/(Si + Zr) ratio ranging from 0 to 1 were synthesized via a coprecipitation method and their physicochemical properties including acid characteristics were investigated. As a result of pyridine adsorbed FT-IR and iso-propanol (IPA)-TPD, the type, quantity, concentration, and strength of acid site were largely dependent on the molar composition of SiO2-ZrO2 mixed oxides, where they reached maximum at the Si/(Si + Zr) molar ratio of 0.7. Furthermore, it was found that the catalytic activity was proportional to the acid strength in the decomposition of IPA, while the quantity of Brønsted acid sites was a principal contributor to the activity in the decomposition of formic acid.



























 京公网安备 11010802027423号
京公网安备 11010802027423号