Science Bulletin ( IF 18.9 ) Pub Date : 2017-11-24 , DOI: 10.1016/j.scib.2017.11.017 Lijie Zhong 1 , Jens Oluf Jensen 1 , Lars Nilausen Cleemann 1 , Chao Pan 1 , Qingfeng Li 1
|
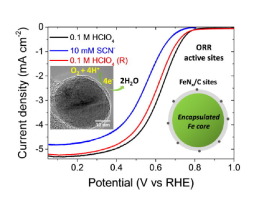
The graphitic-layer encapsulated iron-containing nanoparticles ([email protected]) have been proposed as a potential type of active and stable non-precious metal electrocatalysts (NPMCs) for the oxygen reduction reaction (ORR). However, the contribution of the encapsulated components to the ORR activity is still unclear compared with the well-recognized surface coordinated FeNx/C structure. Using the strong complexing effect of the iron component with anions, cyanide (CN−) in alkaline and thiocyanate (SCN−) in acidic media, the metal containing active sites are electrochemically probed. Three representative catalysts are chosen for a comparison including the as-prepared encapsulated [email protected], commercial Fe/N/C catalyst with iron–nitrogen coordinated surface functionalities and molecular iron phthalocyanine (FePc) containing well-defined structures and compositions. It was found that all samples showed significant shifts of half-wave potentials indicating that surface Fe coordinated sites in all cases. The [email protected] catalyst showed the weakest poisoning effect (the lowest shifts of half-wave potential) compared to the Fe/N/C and FePc catalysts in both electrolytes. These results could be explained that the encapsulated iron components influence the FeNx/C and/or NxC surface functionality.
中文翻译:

石墨层包封铁氧还原反应电催化剂活性位点的电化学研究
石墨层包封的含铁纳米颗粒([电子邮件保护])已被提议作为用于氧还原反应(ORR)的活性和稳定的非贵金属电催化剂(NPMC)的潜在类型。然而,与公认的表面配位 FeN x /C 结构相比,封装组分对 ORR 活性的贡献仍不清楚。利用铁成分与阴离子的强络合作用,碱中的氰化物(CN -)和硫氰酸盐(SCN -) 在酸性介质中,对含有活性位点的金属进行电化学探测。选择三种代表性催化剂进行比较,包括制备好的封装 [电子邮件保护]、具有铁氮配位表面功能的商用 Fe/N/C 催化剂和包含明确结构和组成的分子铁酞菁 (FePc)。发现所有样品都显示出半波电位的显着变化,表明在所有情况下表面 Fe 配位。与两种电解质中的 Fe/N/C 和 FePc 催化剂相比,[email protected] 催化剂表现出最弱的中毒效应(半波电位的最低偏移)。这些结果可以解释为封装的铁组分影响 FeN x /C 和/或 N xC面功能。



























 京公网安备 11010802027423号
京公网安备 11010802027423号