当前位置:
X-MOL 学术
›
Angew. Chem. Int. Ed.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
ortho‐Directing Chromium Arene Complexes as Efficient Mediators for Enantiospecific C(sp2)–C(sp3) Cross‐Coupling Reactions
Angewandte Chemie International Edition ( IF 16.6 ) Pub Date : 2018-01-03 , DOI: 10.1002/anie.201711816 Raphael Bigler 1 , Varinder K. Aggarwal 1
Angewandte Chemie International Edition ( IF 16.6 ) Pub Date : 2018-01-03 , DOI: 10.1002/anie.201711816 Raphael Bigler 1 , Varinder K. Aggarwal 1
Affiliation
|
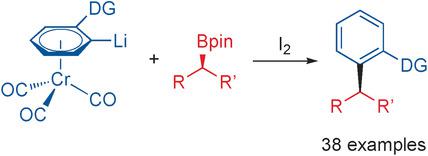
A new strategy for the coupling of a broad scope of electronically diverse aromatics to boronic esters is reported. The coupling sequence, which relies on the directed ortho‐lithiation of chromium arene complexes followed by boronate formation and oxidation, occurs with complete ortho‐selectivity and enantiospecificity to give the coupling products in excellent yields and with high functional group tolerance. An intermediate chromium arene boronate complex was characterized by X‐ray, NMR, and IR experiments to elucidate the reaction mechanism.
中文翻译:

正交定向铬芳烃配合物作为对映体特异性C(sp2)–C(sp3)交叉偶联反应的高效介体
报道了一种新的策略,该策略用于将多种电子多样性的芳族化合物偶联至硼酸酯。偶联顺序依赖于铬芳烃络合物的直接邻位锂化反应,然后形成硼酸酯和氧化反应,具有完全的邻位选择性和对映体特异性,从而以优异的收率和较高的官能团耐受性提供偶联产物。通过X射线,NMR和IR实验对中间体铬芳烃硼酸酯络合物进行了表征,以阐明反应机理。
更新日期:2018-01-03
中文翻译:

正交定向铬芳烃配合物作为对映体特异性C(sp2)–C(sp3)交叉偶联反应的高效介体
报道了一种新的策略,该策略用于将多种电子多样性的芳族化合物偶联至硼酸酯。偶联顺序依赖于铬芳烃络合物的直接邻位锂化反应,然后形成硼酸酯和氧化反应,具有完全的邻位选择性和对映体特异性,从而以优异的收率和较高的官能团耐受性提供偶联产物。通过X射线,NMR和IR实验对中间体铬芳烃硼酸酯络合物进行了表征,以阐明反应机理。


























 京公网安备 11010802027423号
京公网安备 11010802027423号