Bioorganic Chemistry ( IF 5.1 ) Pub Date : 2017-12-02 , DOI: 10.1016/j.bioorg.2017.12.001 Muhammad Taha , Syahrul Imran , Fazal Rahim , Abdul Wadood , Khalid Mohammed Khan
|
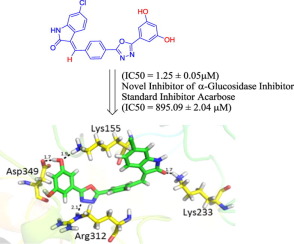
Inhibition of α-glucosidase is an effective strategy for controlling post-prandial hyperglycemia in diabetic patients. Beside these α-glucosidase inhibitors has been also used as anti-obesity and anti-viral drugs. Keeping in view the greater importance of α-glucosidase inhibitors here in this study we are presenting oxindole based oxadiazoles hybrid analogs (1–20) synthesis, characterized by different spectroscopic techniques including 1H NMR and EI-MS and their α-glucosidase inhibitory activity. All compounds were found potent inhibitors for the enzyme with IC50 values ranging between 1.25 ± 0.05 and 268.36 ± 4.22 µM when compared with the standard drug acarbose having IC50 value 895.09 ± 2.04 µM. Our study identifies novel series of potent α-glucosidase inhibitors and further investigation on this may led to the lead compounds. A structure activity relationship has been established for all compounds. The interactions of the active compounds and enzyme active site were established with the help of molecular docking studies.
中文翻译:

基于羟吲哚的恶二唑杂合类似物:新型α-葡萄糖苷酶抑制剂
抑制α-葡萄糖苷酶是控制糖尿病患者餐后高血糖的有效策略。除了这些α-葡萄糖苷酶抑制剂外,还被用作抗肥胖和抗病毒药物。考虑到α-葡萄糖苷酶抑制剂在本研究中的重要性,我们在此介绍基于氧吲哚的恶二唑杂合类似物(1-20)的合成,其特征在于不同的光谱技术,包括1 H NMR和EI-MS及其对α-葡萄糖苷酶的抑制活性。与具有IC的标准药物阿卡波糖相比,所有化合物均被认为是该酶的有效抑制剂,IC 50值在1.25±0.05至268.36±4.22 µM之间。50值895.09±2.04 µM。我们的研究确定了一系列新的有效的α-葡萄糖苷酶抑制剂,对此的进一步研究可能导致了先导化合物的产生。已经为所有化合物建立了结构活性关系。借助分子对接研究建立了活性化合物与酶活性位点之间的相互作用。



























 京公网安备 11010802027423号
京公网安备 11010802027423号