Journal of Fluorine Chemistry ( IF 1.9 ) Pub Date : 2017-05-22 , DOI: 10.1016/j.jfluchem.2017.05.009 Anikó Nemes , Egmont Mérész , István Jalsovszky , Dénes Szabó , Zsolt Böcskei , József Rábai
|
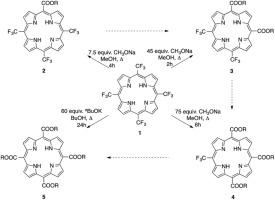
Based on the unusual reactivity of trifluoromethyl groups in nitrogen containing heterocycles, we synthesized the appropriate porphyrin mono-, di-, tri- or tetra-carboxylic ester derivatives by treatment of the precursor meso-tetrakis(trifluoromethyl)porphyrin with an excess of sodium- or potassium alkoxide in the respective alcohol. This method offers an efficient route for the synthesis of lower symmetry meso-substituted porphyrins compared to usual preparations utilizing stepwise condensation reactions. The structure of tetrakis(butyloxycarbonyl)porphyrin 5 was determined by X-ray analysis.
中文翻译:

在三氟甲基基团的不寻常的反应性内消旋-四(三氟甲基)卟啉
基于三氟甲基基团的含氮杂环的不寻常的反应性,我们合成了相应的卟啉单,二,三或四羧酸酯衍生物通过处理前体的内消旋-四(三氟甲基)卟啉与过量的钠或相应醇中的烷氧基钾。与使用逐步缩合反应的常规制备方法相比,该方法为合成较低对称的内消旋取代的卟啉提供了一条有效途径。通过X射线分析确定四(丁氧基羰基)卟啉5的结构。



























 京公网安备 11010802027423号
京公网安备 11010802027423号