Catalysis Today ( IF 5.3 ) Pub Date : 2017-11-21 , DOI: 10.1016/j.cattod.2017.11.025 Katarzyna Czyzewska , Anna Trusek-Holownia , Magdalena Dabrowa , Felipe Sarmiento , Jenny M. Blamey
|
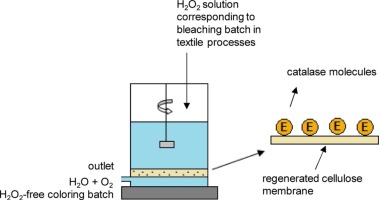
The present work describes the results of the preliminary characterization of a novel enzyme with catalase activity isolated from psychrotolerant bacteria. Catalases decompose hydrogen peroxide (H2O2) to water and oxygen. Chemical immobilization is the best solution in case of reactions in which a gaseous product is created hence immobilization via divinyl sulfone having used a polymeric regenerated cellulose membrane was proposed. The preparations were checked according to their activity and stability.
In the textile industry, a catalase is used to decompose the hydrogen peroxide waste after bleaching fabrics. During photocatalytic oxidation, H2O2 is added at relatively high concentration app. 20–80 g/L and the pH of these solutions is in the range between 7 and 10.
The psychrophilic catalase described in this work presents high activity up to 40 g/L at pH 7–10, while a commercial alternative isolated from bovine liver only displays activity up to 14 g/L at pH not higher than 9. The stability of enzymatic activity is strongly dependent on temperature and is also a function of substrate concentration.
The results obtained with this psychrophilic catalase are promising for textile effluence treatment.
中文翻译:

用于H 2 O 2分解的催化膜
本工作描述了从精神抗性细菌中分离出的具有过氧化氢酶活性的新型酶的初步表征结果。过氧化氢酶将过氧化氢(H 2 O 2)分解为水和氧气。在产生气态产物的反应的情况下,化学固定化是最好的解决方案,因此提出了通过使用聚合物再生的纤维素膜的二乙烯基砜进行固定化的反应。根据其活性和稳定性检查制剂。
在纺织工业中,过氧化氢酶用于在漂白织物后分解过氧化氢废物。在光催化氧化期间,以相对较高的浓度app添加H 2 O 2。20–80 g / L,这些溶液的pH值在7到10之间。
这项工作中描述的嗜冷性过氧化氢酶在pH 7-10时具有高达40 g / L的高活性,而从牛肝中分离出的商业替代品在pH不高于9时仅具有14 g / L的活性。活性很大程度上取决于温度,并且也是底物浓度的函数。
用该嗜冷性过氧化氢酶获得的结果有望用于纺织品废水处理。


























 京公网安备 11010802027423号
京公网安备 11010802027423号