Catalysis Today ( IF 5.3 ) Pub Date : 2017-11-21 , DOI: 10.1016/j.cattod.2017.11.022 Jhulimar M. Celedonio , Rose Mardie Pacia , Young Soo Ko
|
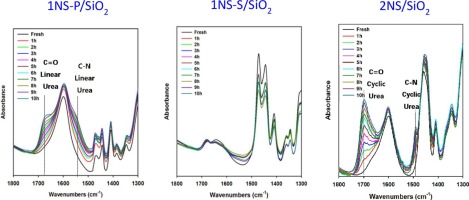
Since the development of amine-functionalized adsorbent is critical for its large-scale implementation, an investigation on the stability in realistic process condition is significantly important. The amine-functionalized CO2 adsorbents were prepared via impregnation of primary amine (1NS-P/SiO2), secondary amine (1NS-S/SiO2) and diamines (2NS/SiO2) into a silica support. The degradation products that could have caused the deactivation of the adsorbents are determined and quantified by in-situ FT-IR measurement and deconvoluted FT-IR spectra. The adsorbents were evaluated for their stability in multiple temperature swing adsorption (TSA) cycles and long-time CO2 or air exposure at elevated temperature. It revealed that the degradation of adsorbents and its rate depended on the amine structure and gas conditions and that two main degradation species were urea and amide. Primary amine was more prone to CO2-induced degradation than secondary amine. Secondary amine was less stable than primary amine for O2-induced degradation. Diamine showed both CO2- and O2-induced degradations. To assess which has a more detrimental effect on the stability of the adsorbents between CO2 and O2 gases, the effect of long-time exposure of the adsorbents in pure CO2 or air was determined at 150 °C using both TG and in-situ FT-IR measurement. The long-time exposure of 1NS-P/SiO2 to CO2 caused about 50% loss in CO2 uptake with the faster accumulation of linear urea. The long-time exposure to air caused just about 13% loss in CO2 uptake with the slower accumulation of amide. 1NS-S/SiO2 showed better stability for CO2 than 1NS-P/SiO2 without any decrease in capacity and without any changes in its spectra. 2NS/SiO2 showed that urea formed faster than amide. It was degraded more in air than in CO2 with about 92% and 51% loss of CO2 uptake, respectively. It can be inferred from the results that O2-induced degradation is more detrimental than CO2-induced degradation.
中文翻译:

烟气条件下CO 2吸附剂的多环和长期稳定性的光谱研究
由于胺官能化吸附剂的开发对其大规模实施至关重要,因此研究实际工艺条件下的稳定性非常重要。该胺官能化的CO 2吸附剂是通过伯胺的浸渍(1NS-P /制备的SiO 2),仲胺(1NS-S /的SiO 2)和二胺(2NS /二氧化硅2)到二氧化硅载体上。通过原位FT-IR测量和去卷积的FT-IR光谱确定并量化可能引起吸附剂失活的降解产物。评价了吸附剂在多个变温吸附(TSA)循环和长时间CO 2中的稳定性或在高温下暴露于空气中。结果表明,吸附剂的降解及其速率取决于胺的结构和气体条件,两种主要的降解物质是尿素和酰胺。伯胺比仲胺更容易发生CO 2诱导的降解。对于O 2诱导的降解,仲胺的稳定性不如伯胺。二胺显示出CO 2和O 2诱导的降解。为了评估哪种吸附剂对CO 2和O 2气体之间的吸附剂的稳定性具有更有害的影响,即吸附剂长时间暴露于纯CO 2中的影响或使用TG和原位FT-IR测量在150°C下确定空气。1NS-P / SiO 2长时间暴露于CO 2导致线性尿素积累更快,CO 2吸收损失约50%。长时间暴露在空气中会导致酰胺吸收缓慢,导致CO 2吸收量减少约13%。1NS-S / SiO 2对CO 2的稳定性优于1NS-P / SiO 2,而容量没有下降,光谱也没有变化。2NS / SiO 2表明,尿素的形成速度快于酰胺。它被降解更在空气中比在CO 2与CO的约92%和51%的损失2摄取。从结果可以推断,O 2诱导的降解比CO 2诱导的降解更有害。

























 京公网安备 11010802027423号
京公网安备 11010802027423号