当前位置:
X-MOL 学术
›
J. Anal. At. Spectrom.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Evaluation of approaches to the abatement of nitrate interference with photochemical vapor generation
Journal of Analytical Atomic Spectrometry ( IF 3.4 ) Pub Date : 2017-10-27 00:00:00 , DOI: 10.1039/c7ja00311k Gisele S. Lopes 1, 2, 3, 4 , Ralph E. Sturgeon 5, 6, 7, 8 , Patricia Grinberg 5, 6, 7, 8 , Enea Pagliano 5, 6, 7, 8
Journal of Analytical Atomic Spectrometry ( IF 3.4 ) Pub Date : 2017-10-27 00:00:00 , DOI: 10.1039/c7ja00311k Gisele S. Lopes 1, 2, 3, 4 , Ralph E. Sturgeon 5, 6, 7, 8 , Patricia Grinberg 5, 6, 7, 8 , Enea Pagliano 5, 6, 7, 8
Affiliation
|
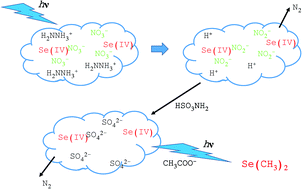
Photochemical vapor generation (PVG) provides an alternative to chemical vapor generation techniques but suffers severe interference in the presence of oxidizing species, notably nitrate and nitrite. Although the mechanism is not completely understood, scavenging of photogenerated free electrons and hydrogen radicals by NO2− and NO3− likely plays a role in terminating analyte reduction reactions which lead to hydridization, carbonylation or alkylation of analytes. In this study, several strategies for the elimination of nitrate and nitrite from samples prior to PVG were examined. Since the Se(IV)-acetic acid PVG system proved to be extremely sensitive to low levels of these oxidants, wherein 50% loss of generation efficiency is realized at [NO3−] > 2.5 mM, it was used as a test case. To this end, a number of practical approaches to the reduction of nitrate and nitrite were evaluated, including: utilizing sulfamic acid, hydrazine sulfate, sodium thiosulfate and V(III), as well as removal by photo-reduction and quantitative precipitation by nitron. Whereas none of these approaches could be implemented with impunity, a methodology based on UV photolysis of a basic solution containing 2% hydrazine sulfate provided high NO3− removal efficiencies. Residual NO2− was quantitatively eliminated by addition of sulfamic acid to the acidified solution, allowing efficient generation of volatile Se(CH3)2 species. The methodology was tested by application to Certified Reference Material SLRS-3 (river water, acidified to pH 1.6 with nitric acid), achieving 83 ± 2% Se(IV) spike recoveries using PVG sample introduction coupled with ICP-MS for detection, illustrating effective control over interferences and recovery of Se(IV).
中文翻译:

消除硝酸盐对光化学蒸气产生的干扰的方法的评价
光化学蒸气产生(PVG)提供了化学蒸气产生技术的替代方法,但在存在氧化性物质(尤其是硝酸盐和亚硝酸盐)的情况下受到严重干扰。虽然机理还不完全清楚,通过NO清除光生的自由电子和氢自由基的2 -和NO 3 -可能起着终止分析物还原反应,其导致hydridization,羰基化或分析物的烷基化作用。在这项研究中,研究了几种在PVG之前从样品中消除硝酸盐和亚硝酸盐的策略。自Se(IV)-乙酸PVG系统被证明对这些氧化剂的低含量极为敏感,在[NO 3 - ]> 2.5 mM时实现了50%的发电效率损失,它被用作测试案例。为此,评估了许多减少硝酸盐和亚硝酸盐的实用方法,包括:利用氨基磺酸,硫酸肼,硫代硫酸钠和V(III),以及通过光还原除去和通过硝化定量沉淀。而这些方法都没有可以不受惩罚来实现,基于UV的方法光解含有高提供NO 2%硫酸肼的碱性溶液的3 -去除效率。剩余NO 2 -通过将氨基磺酸添加到酸化溶液中来定量地消除汞,从而可以有效地产生挥发性的Se(CH 3)2物质。该方法已通过应用于认证参考材料SLRS-3(河水,用硝酸酸化至pH 1.6)进行了测试,使用PVG样品引入与ICP-MS结合检测达到了83±2%的Se(IV)加标回收率,说明了有效控制Se(IV)的干扰和恢复。
更新日期:2017-10-27
中文翻译:

消除硝酸盐对光化学蒸气产生的干扰的方法的评价
光化学蒸气产生(PVG)提供了化学蒸气产生技术的替代方法,但在存在氧化性物质(尤其是硝酸盐和亚硝酸盐)的情况下受到严重干扰。虽然机理还不完全清楚,通过NO清除光生的自由电子和氢自由基的2 -和NO 3 -可能起着终止分析物还原反应,其导致hydridization,羰基化或分析物的烷基化作用。在这项研究中,研究了几种在PVG之前从样品中消除硝酸盐和亚硝酸盐的策略。自Se(IV)-乙酸PVG系统被证明对这些氧化剂的低含量极为敏感,在[NO 3 - ]> 2.5 mM时实现了50%的发电效率损失,它被用作测试案例。为此,评估了许多减少硝酸盐和亚硝酸盐的实用方法,包括:利用氨基磺酸,硫酸肼,硫代硫酸钠和V(III),以及通过光还原除去和通过硝化定量沉淀。而这些方法都没有可以不受惩罚来实现,基于UV的方法光解含有高提供NO 2%硫酸肼的碱性溶液的3 -去除效率。剩余NO 2 -通过将氨基磺酸添加到酸化溶液中来定量地消除汞,从而可以有效地产生挥发性的Se(CH 3)2物质。该方法已通过应用于认证参考材料SLRS-3(河水,用硝酸酸化至pH 1.6)进行了测试,使用PVG样品引入与ICP-MS结合检测达到了83±2%的Se(IV)加标回收率,说明了有效控制Se(IV)的干扰和恢复。

























 京公网安备 11010802027423号
京公网安备 11010802027423号