当前位置:
X-MOL 学术
›
Adv. Energy Mater.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Reducing Mg Anode Overpotential via Ion Conductive Surface Layer Formation by Iodine Additive
Advanced Energy Materials ( IF 27.8 ) Pub Date : 2017-10-24 , DOI: 10.1002/aenm.201701728 Xiaogang Li 1, 2 , Tao Gao 1 , Fudong Han 1 , Zhaohui Ma 1 , Xiulin Fan 1 , Singyuk Hou 1 , Nico Eidson 1 , Weishan Li 2 , Chunsheng Wang 1
Advanced Energy Materials ( IF 27.8 ) Pub Date : 2017-10-24 , DOI: 10.1002/aenm.201701728 Xiaogang Li 1, 2 , Tao Gao 1 , Fudong Han 1 , Zhaohui Ma 1 , Xiulin Fan 1 , Singyuk Hou 1 , Nico Eidson 1 , Weishan Li 2 , Chunsheng Wang 1
Affiliation
|
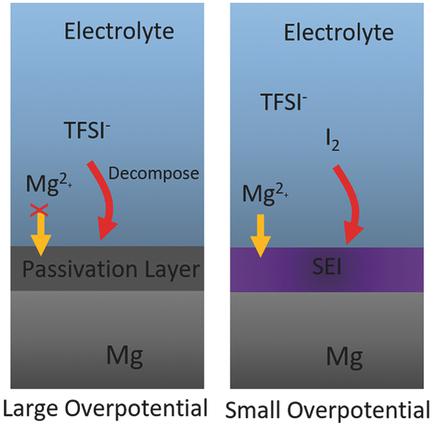
Electrolytes that are able to reversibly deposit/strip Mg are crucial for rechargeable Mg batteries. The most studied complex electrolytes based on Lewis acid‐base chemistry are expensive, difficult to be synthesized, and show limited anodic stability. Conventional electrolytes using simple salts such as Mg(TFSI)2 can be readily synthesized, but Mg deposition/stripping in these simple salt electrolytes is accompanied by a large overpotential due to the formation of a surface layer on the Mg metal with a low Mg ion conductivity. Here the overpotential for Mg deposition/stripping in a simple salt, Mg(TFSI)2‐1,2‐dimethoxyethane (DME), electrolyte is significantly reduced by adding a small concentration of iodine (≤50 × 10−3m) as an additive. Mechanism studies demonstrate that an Mg ion conductive solid MgI2 layer is formed on the surface of the Mg metal and acts as a solid electrolyte interface. With the Mg(TFSI)2‐DME‐I2 electrolyte, a very small voltage hysteresis is achieved in an Mg‐S full cell.
中文翻译:

通过碘添加剂通过离子导电表面层的形成减少镁阳极过电势
能够可逆地沉积/剥离Mg的电解质对于可充电Mg电池至关重要。研究最多的基于路易斯酸碱化学的复杂电解质价格昂贵,难以合成,并且显示出有限的阳极稳定性。可以容易地合成使用诸如Mg(TFSI)2之类的简单盐的常规电解质,但是由于在具有低Mg离子的Mg金属上形成表面层,因此在这些简单盐电解质中的Mg沉积/剥离伴随着较大的超电势。电导率。这里过电位镁沉积/剥离以简单的盐,镁(TFSI)2 -1,2-二甲氧基乙烷(DME),电解质被显著加入碘的小浓度(≤50×10降低-3米)作为添加剂。机理研究表明,Mg离子传导性固体MgI 2层形成在Mg金属的表面上,并充当固体电解质界面。使用Mg(TFSI)2 -DME-I 2电解质,在Mg-S满电池中可实现非常小的电压滞后。
更新日期:2017-10-24
中文翻译:

通过碘添加剂通过离子导电表面层的形成减少镁阳极过电势
能够可逆地沉积/剥离Mg的电解质对于可充电Mg电池至关重要。研究最多的基于路易斯酸碱化学的复杂电解质价格昂贵,难以合成,并且显示出有限的阳极稳定性。可以容易地合成使用诸如Mg(TFSI)2之类的简单盐的常规电解质,但是由于在具有低Mg离子的Mg金属上形成表面层,因此在这些简单盐电解质中的Mg沉积/剥离伴随着较大的超电势。电导率。这里过电位镁沉积/剥离以简单的盐,镁(TFSI)2 -1,2-二甲氧基乙烷(DME),电解质被显著加入碘的小浓度(≤50×10降低-3米)作为添加剂。机理研究表明,Mg离子传导性固体MgI 2层形成在Mg金属的表面上,并充当固体电解质界面。使用Mg(TFSI)2 -DME-I 2电解质,在Mg-S满电池中可实现非常小的电压滞后。

























 京公网安备 11010802027423号
京公网安备 11010802027423号