当前位置:
X-MOL 学术
›
Org. Chem. Front.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
1,2,3-Triazole-assisted C–H amidation by cobalt(III) catalysis
Organic Chemistry Frontiers ( IF 5.4 ) Pub Date : 2017-08-10 00:00:00 , DOI: 10.1039/c7qo00556c Xinling Yu 1, 2, 3, 4, 5 , Qiang Ma 1, 2, 3, 4, 5 , Songyang Lv 1, 2, 3, 4, 5 , Jue Li 1, 2, 3, 4, 5 , Chen Zhang 1, 2, 3, 4, 5 , Li Hai 1, 2, 3, 4, 5 , Qiantao Wang 1, 2, 3, 4, 5 , Yong Wu 1, 2, 3, 4, 5
Organic Chemistry Frontiers ( IF 5.4 ) Pub Date : 2017-08-10 00:00:00 , DOI: 10.1039/c7qo00556c Xinling Yu 1, 2, 3, 4, 5 , Qiang Ma 1, 2, 3, 4, 5 , Songyang Lv 1, 2, 3, 4, 5 , Jue Li 1, 2, 3, 4, 5 , Chen Zhang 1, 2, 3, 4, 5 , Li Hai 1, 2, 3, 4, 5 , Qiantao Wang 1, 2, 3, 4, 5 , Yong Wu 1, 2, 3, 4, 5
Affiliation
|
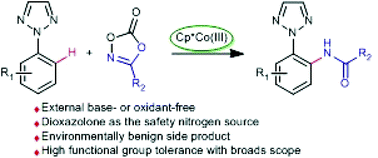
A procedure for the [Cp*CoIII]-catalyzed, 1,2,3-triazole directed C–H amidation of arenes, using dioxazolone as an efficient amidating reagent, has been developed. This reaction provides a step- and atom-economical protocol for C–N bond formation, releasing carbon dioxide as a single byproduct. Yet a variety of functional groups are well tolerated, obtaining the desired products in moderate to excellent yields.
中文翻译:

钴(III)催化1,2,3-三唑辅助的CH酰胺化
已经开发了一种使用二恶唑酮作为有效酰胺化试剂的[Cp * Co III ]催化的1,2,3-三唑直接催化芳烃的CH酰胺化反应的方法。该反应为C–N键的形成提供了一步和原子经济的方法,从而释放出二氧化碳作为单一副产物。然而,各种官能团的耐受性很好,以中等至极好的收率获得了所需的产物。
更新日期:2017-10-25
中文翻译:

钴(III)催化1,2,3-三唑辅助的CH酰胺化
已经开发了一种使用二恶唑酮作为有效酰胺化试剂的[Cp * Co III ]催化的1,2,3-三唑直接催化芳烃的CH酰胺化反应的方法。该反应为C–N键的形成提供了一步和原子经济的方法,从而释放出二氧化碳作为单一副产物。然而,各种官能团的耐受性很好,以中等至极好的收率获得了所需的产物。

























 京公网安备 11010802027423号
京公网安备 11010802027423号