当前位置:
X-MOL 学术
›
Org. Lett.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
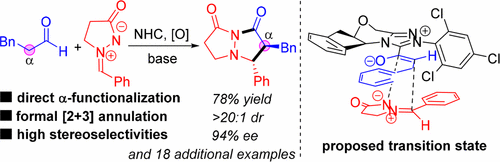
Oxidative Asymmetric [2 + 3] Annulation of Aldehydes with Azomethine Imines Enabled by N-Heterocyclic Carbene Catalysis
Organic Letters ( IF 5.2 ) Pub Date : 2017-11-02 00:00:00 , DOI: 10.1021/acs.orglett.7b02948 Shiru Yuan 1 , Yuchen Luo 1 , Jingyi Peng 1 , Maozhong Miao 1 , Jianfeng Xu 1 , Hongjun Ren 1
Organic Letters ( IF 5.2 ) Pub Date : 2017-11-02 00:00:00 , DOI: 10.1021/acs.orglett.7b02948 Shiru Yuan 1 , Yuchen Luo 1 , Jingyi Peng 1 , Maozhong Miao 1 , Jianfeng Xu 1 , Hongjun Ren 1
Affiliation
|
An efficient asymmetric [2 + 3] annulation of simple aldehydes with N,N′-cyclic azomethine imines via oxidative N-heterocyclic carbene (NHC) catalysis is disclosed, affording bicyclic pyrazolidinones bearing two vicinal tertiary stereogenic centers with moderate to good yields (56–84% for 19 examples), good to excellent diastereoselectivities (>20:1 for 17 examples), and good to excellent enantioselectivities (66–98% for 19 examples). This direct α-carbon functionalization reaction of aldehyde can be readily performed on gram scale under mild conditions, and a five-membered transition state is proposed to rationalize the stereochemical outcome.
中文翻译:

N-杂环卡宾催化实现的具有亚甲亚胺的醛的氧化不对称[2 + 3]环化
揭示了通过氧化N-杂环卡宾(NHC)催化与N,N'-环偶氮甲亚胺的简单醛类的有效不对称[2 + 3]环合反应,提供了带有两个邻近的三级立体立体中心且产率中等至良好的双环吡唑烷酮(56) – 19%的样品为–84%),非对映选择性好至极好(17个实例> 20:1),对映体选择性好至极好(19个实例为66–98%)。醛的这种直接α-碳官能化反应可在温和条件下轻松以克为单位进行,并提出了五元过渡态以使立体化学结果合理化。
更新日期:2017-11-02
中文翻译:

N-杂环卡宾催化实现的具有亚甲亚胺的醛的氧化不对称[2 + 3]环化
揭示了通过氧化N-杂环卡宾(NHC)催化与N,N'-环偶氮甲亚胺的简单醛类的有效不对称[2 + 3]环合反应,提供了带有两个邻近的三级立体立体中心且产率中等至良好的双环吡唑烷酮(56) – 19%的样品为–84%),非对映选择性好至极好(17个实例> 20:1),对映体选择性好至极好(19个实例为66–98%)。醛的这种直接α-碳官能化反应可在温和条件下轻松以克为单位进行,并提出了五元过渡态以使立体化学结果合理化。

























 京公网安备 11010802027423号
京公网安备 11010802027423号