Tetrahedron ( IF 2.1 ) Pub Date : 2017-09-18 , DOI: 10.1016/j.tet.2017.09.023 Lidiya Fedenok , Fedor Dultsev , Igor Barabanov , Nikolay Polyakov
|
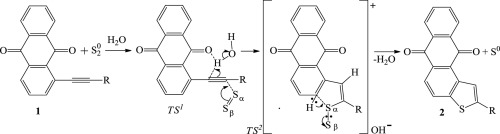
The CH-cyclization of alkynyl-9,10-anthraquinones 1 into thienoanthraquinones 2 is shown to be initiated by the neutral S2 molecule and not by the sulfide anion as it is assumed in the existing model of CH-cyclization with the nucleophiolic substitution of H. The formation of S2 takes place in the reaction medium due to the partial reduction of the substrate 1 by Na2S into the corresponding anthrahydroquinone, which is inactive in the main reaction. We present a new cyclization mechanism with the key stage involving the electrophilic displacement of H, which results in the formation of the reaction product 2 and elimination of S. In the reaction medium atomic sulfur is transformed into S2, which initiates the next cyclization act. This determines the chain nature of cyclization with the participation of S2. Within this model, the route to increase the yield of target thienoanthraquinones is proposed through the introduction of a neutral quinone able to generate S2 by means of easy reduction.
中文翻译:

硫化钠参与的炔基蒽醌С-Н环化成硫杂环蒽醌的机理
炔基-9,10-蒽醌的CH-环化1到thienoanthraquinones 2被示出为通过中性S为发起2分子,不是如假定在CH-环化的现有模型的nucleophiolic取代的硫化物阴离子H. S 2的形成在反应介质中发生,这是因为底物1被Na 2 S部分还原成相应的蒽氢醌,而后者在主反应中没有活性。我们提出了一种新的环化机理,其关键阶段涉及H的亲电置换,这导致了反应产物2的形成。在反应介质中,原子硫被转化为S 2,从而引发下一个环化作用。这决定了在S 2的参与下环化的链性质。在该模型中,通过引入能够通过简单还原生成S 2的中性醌,提出了提高目标硫代蒽蒽醌产率的途径。


























 京公网安备 11010802027423号
京公网安备 11010802027423号