Bioorganic & Medicinal Chemistry Letters ( IF 2.7 ) Pub Date : 2017-09-06 , DOI: 10.1016/j.bmcl.2017.09.003 Arun K. Ghosh , W. Sean Fyvie , Margherita Brindisi , Melinda Steffey , Johnson Agniswamy , Yuan-Fang Wang , Manabu Aoki , Masayuki Amano , Irene T. Weber , Hiroaki Mitsuya
|
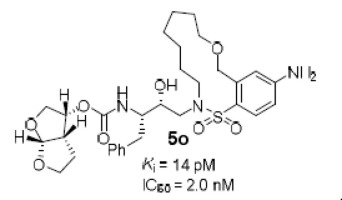
Design, synthesis, and evaluation of a new class of HIV-1 protease inhibitors containing diverse flexible macrocyclic P1′-P2′ tethers are reported. Inhibitor 5a with a pyrrolidinone-derived macrocycle exhibited favorable enzyme inhibitory and antiviral activity (Ki = 13.2 nM, IC50 = 22 nM). Further incorporation of heteroatoms in the macrocyclic skeleton provided macrocyclic inhibitors 5m and 5o. These compounds showed excellent HIV-1 protease inhibitory (Ki = 62 pM and 14 pM, respectively) and antiviral activity (IC50 = 5.3 nM and 2.0 nM, respectively). Inhibitor 5o also remained highly potent against a DRV-resistant HIV-1 variant.
中文翻译:

设计,合成,X射线研究和涉及P1'-P2'配体的新型大环HIV-1蛋白酶抑制剂的生物学评估
报道了设计,合成和评估一类新型的HIV-1蛋白酶抑制剂,其包含多种柔性大环P1'-P2'系链。具有吡咯烷酮衍生的大环的抑制剂5a表现出良好的酶抑制和抗病毒活性(K i = 13.2 nM,IC 50 = 22 nM)。在大环骨架中进一步掺入杂原子可提供大环抑制剂5m和5o。这些化合物显示出出色的HIV-1蛋白酶抑制性(分别为K i = 62 pM和14 pM)和抗病毒活性(IC 50分别 为5.3 nM和2.0 nM)。抑制剂5o 它还对DRV耐药的HIV-1变异株具有很高的效力。



























 京公网安备 11010802027423号
京公网安备 11010802027423号