当前位置:
X-MOL 学术
›
Chem. Commun.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Copper-catalyzed trifluoromethylazidation and rearrangement of aniline-linked 1,7-enynes: access to CF3-substituted azaspirocyclic dihydroquinolin-2-ones and furoindolines
Chemical Communications ( IF 4.9 ) Pub Date : 2017-07-17 00:00:00 , DOI: 10.1039/c7cc04748g Liu-Zhu Yu 1, 2, 3, 4, 5 , Yin Wei 6, 7, 8, 9, 10 , Min Shi 1, 2, 3, 4, 5
Chemical Communications ( IF 4.9 ) Pub Date : 2017-07-17 00:00:00 , DOI: 10.1039/c7cc04748g Liu-Zhu Yu 1, 2, 3, 4, 5 , Yin Wei 6, 7, 8, 9, 10 , Min Shi 1, 2, 3, 4, 5
Affiliation
|
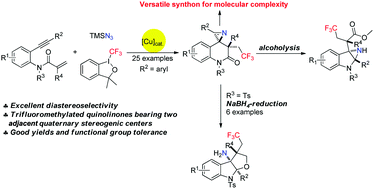
A set of reactions involving copper-catalyzed trifluoromethylazidation and then rearrangement of aniline-linked 1,7-enynes with the relatively poorly reactive Togni reagent I and TMSN3 was developed, and provided facile access to structurally diverse and useful CF3-substituted azaspirocyclic dihydroquinolin-2-ones bearing two adjacent quaternary carbon centers. We were also able to obtain these products on a large scale. Moreover, the obtained products were further transformed into a range of synthetically valuable furoindolines bearing three consecutive quaternary carbon centers after reduction by NaBH4. Also, the delivery of multi-functionalized aziridines as a result of alcoholysis and LiAlH4-reduction of corresponding dihydroquinolin-2-ones 2ba demonstrated the synthetic value of this newly developed protocol.
中文翻译:

铜催化的三氟甲基叠氮化和苯胺连接的1,7-烯炔的重排:获得CF 3取代的氮杂螺环二氢喹啉-2-酮和呋喃二氢吲哚
开发了一套反应,涉及铜催化的三氟甲基叠氮化,然后用相对较差的反应性Togni试剂I和TMSN 3进行苯胺连接的1,7-烯炔的重排,并提供了易于获得的结构多样且有用的CF 3取代的氮杂螺环二氢喹啉的途径。带有两个相邻的季碳中心的-2-ones。我们也能够大规模获得这些产品。此外,将获得的产物在被NaBH 4还原后,进一步转化为一系列具有三个连续的季碳中心的具有合成价值的呋喃二氢吲哚。另外,由于醇解和LiAlH 4的作用,提供了多功能化的氮丙啶-还原相应的二氢喹啉-2-酮2ba证明了该新开发方案的合成价值。
更新日期:2017-07-28
中文翻译:

铜催化的三氟甲基叠氮化和苯胺连接的1,7-烯炔的重排:获得CF 3取代的氮杂螺环二氢喹啉-2-酮和呋喃二氢吲哚
开发了一套反应,涉及铜催化的三氟甲基叠氮化,然后用相对较差的反应性Togni试剂I和TMSN 3进行苯胺连接的1,7-烯炔的重排,并提供了易于获得的结构多样且有用的CF 3取代的氮杂螺环二氢喹啉的途径。带有两个相邻的季碳中心的-2-ones。我们也能够大规模获得这些产品。此外,将获得的产物在被NaBH 4还原后,进一步转化为一系列具有三个连续的季碳中心的具有合成价值的呋喃二氢吲哚。另外,由于醇解和LiAlH 4的作用,提供了多功能化的氮丙啶-还原相应的二氢喹啉-2-酮2ba证明了该新开发方案的合成价值。



























 京公网安备 11010802027423号
京公网安备 11010802027423号