Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Nanolock–Nanopore Facilitated Digital Diagnostics of Cancer Driver Mutation in Tumor Tissue
ACS Sensors ( IF 8.9 ) Pub Date : 2017-07-05 00:00:00 , DOI: 10.1021/acssensors.7b00235 Yong Wang , Kai Tian , Ruicheng Shi , Amy Gu , Michael Pennella , Lindsey Alberts , Kent S. Gates , Guangfu Li , Hongxin Fan 1 , Michael X. Wang , Li-Qun Gu
ACS Sensors ( IF 8.9 ) Pub Date : 2017-07-05 00:00:00 , DOI: 10.1021/acssensors.7b00235 Yong Wang , Kai Tian , Ruicheng Shi , Amy Gu , Michael Pennella , Lindsey Alberts , Kent S. Gates , Guangfu Li , Hongxin Fan 1 , Michael X. Wang , Li-Qun Gu
Affiliation
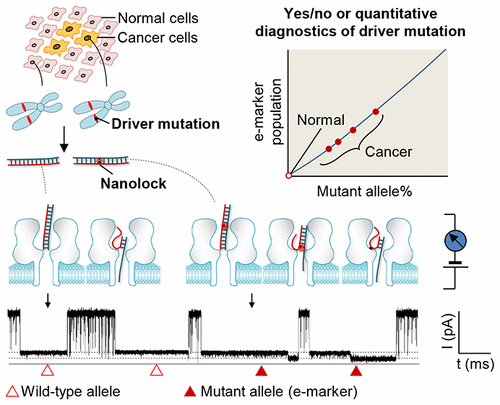
|
Cancer driver mutations are clinically significant biomarkers. In precision medicine, accurate detection of these oncogenic changes in patients would enable early diagnostics of cancer, individually tailored targeted therapy, and precise monitoring of treatment response. Here we investigated a novel nanolock–nanopore method for single-molecule detection of a serine/threonine protein kinase gene BRAF V600E mutation in tumor tissues of thyroid cancer patients. The method lies in a noncovalent, mutation sequence-specific nanolock. We found that the nanolock formed on the mutant allele/probe duplex can separate the duplex dehybridization procedure into two sequential steps in the nanopore. Remarkably, this stepwise unzipping kinetics can produce a unique nanopore electric marker, with which a single DNA molecule of the cancer mutant allele can be unmistakably identified in various backgrounds of the normal wild-type allele. The single-molecule sensitivity for mutant allele enables both binary diagnostics and quantitative analysis of mutation occurrence. In the current configuration, the method can detect the BRAF V600E mutant DNA lower than 1% in the tumor tissues. The nanolock–nanopore method can be adapted to detect a broad spectrum of both transversion and transition DNA mutations, with applications from diagnostics to targeted therapy.
中文翻译:

Nanolock–Nanopore促进肿瘤组织中癌症驱动基因突变的数字诊断
癌症驱动基因突变是临床上重要的生物标志物。在精密医学中,准确检测患者的这些致癌变化将有助于癌症的早期诊断,个性化的靶向治疗以及对治疗反应的精确监测。在这里,我们研究了一种新颖的纳锁-纳米孔方法,用于单分子检测丝氨酸/苏氨酸蛋白激酶基因BRAF甲状腺癌患者肿瘤组织中的V600E突变。该方法在于非共价的,突变序列特异性的纳米锁。我们发现,在突变等位基因/探针双链体上形成的纳米锁可以将双链体去杂化程序分成纳米孔中的两个连续步骤。值得注意的是,这种逐步的解链动力学可以产生独特的纳米孔电标记,利用该标记可以在正常野生型等位基因的各种背景中毫无疑问地鉴定出癌症突变体等位基因的单个DNA分子。对突变型等位基因的单分子敏感性可以对突变发生情况进行二进制诊断和定量分析。在当前配置下,该方法可以检测到BRAF肿瘤组织中的V600E突变型DNA低于1%。纳米锁定-纳米孔方法可适用于检测从诊断到靶向治疗的广泛范围的转化和过渡DNA突变。
更新日期:2017-07-05
中文翻译:

Nanolock–Nanopore促进肿瘤组织中癌症驱动基因突变的数字诊断
癌症驱动基因突变是临床上重要的生物标志物。在精密医学中,准确检测患者的这些致癌变化将有助于癌症的早期诊断,个性化的靶向治疗以及对治疗反应的精确监测。在这里,我们研究了一种新颖的纳锁-纳米孔方法,用于单分子检测丝氨酸/苏氨酸蛋白激酶基因BRAF甲状腺癌患者肿瘤组织中的V600E突变。该方法在于非共价的,突变序列特异性的纳米锁。我们发现,在突变等位基因/探针双链体上形成的纳米锁可以将双链体去杂化程序分成纳米孔中的两个连续步骤。值得注意的是,这种逐步的解链动力学可以产生独特的纳米孔电标记,利用该标记可以在正常野生型等位基因的各种背景中毫无疑问地鉴定出癌症突变体等位基因的单个DNA分子。对突变型等位基因的单分子敏感性可以对突变发生情况进行二进制诊断和定量分析。在当前配置下,该方法可以检测到BRAF肿瘤组织中的V600E突变型DNA低于1%。纳米锁定-纳米孔方法可适用于检测从诊断到靶向治疗的广泛范围的转化和过渡DNA突变。

























 京公网安备 11010802027423号
京公网安备 11010802027423号